Exogenous Ca2+-ATPase isoform effects on Ca2+ transients of embryonic chicken and neonatal rat cardiac myocytes
- PMID: 11018105
- PMCID: PMC2270107
- DOI: 10.1111/j.1469-7793.2000.00053.x
Exogenous Ca2+-ATPase isoform effects on Ca2+ transients of embryonic chicken and neonatal rat cardiac myocytes
Abstract
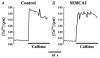
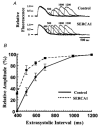
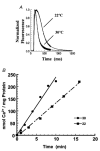
1. Sarco-endoplasmic reticulum Ca2+-ATPase from fast skeletal (SERCA1) or cardiac muscle (SERCA2a) was expressed in embryonic chicken and neonatal rat cardiac myocytes by adenovirus vectors, with c-myc tags on both constructs to compare expression and distinguish exogenous from endogenous SERCA2a in myocytes. 2. Expression of the two isoforms was similar (approximately 3-fold higher than endogenous SERCA). However, SERCA1 activity was 2-fold greater than SERCA2a activity, due to intrinsic differences in turnover rates. Activation of both exogenous SERCA isoforms by Ca2+ was displaced to slightly lower [Ca2+], suggesting that the overexpressed isoforms were independent of phospholamban. In fact, phospholamban and calsequestrin expression were unchanged. 3. Decay time constants of cytosolic Ca2+ transients from cells overexpressing SERCA1 were reduced by 30-40 % and half-widths by 10-15 % compared to controls. SERCA2a overexpression produced much less acceleration of transients in chick than in rat, and less acceleration than SERCA1 overexpression in either species. There was no significant change in resting [Ca2+], peak amplitudes, or in the amount of Ca2+ releasable by caffeine from overexpression of either SERCA isoform. However, the amplitudes of the transients increased with SERCA1 overexpression when pacing frequency limited refilling of the sarcoplasmic reticulum. 4. It is concluded that total SERCA transport velocity has a primary effect on the decay phase of transients. Transport velocity is affected by SERCA isoform turnover rate, temperature, and/or SERCA copy number.
Figures




References
-
- Anger M, Samuel JL, Marotte F, Wuytack F, Rappaport L, Lompre AM. In situ mRNA distribution of sarco(endo)plasmic reticulum Ca(2+)-ATPase isoforms during ontogeny in the rat. Journal of Molecular and Cellular Cardiology. 1994;26:539–550. - PubMed
-
- Baker DL, Hashimoto K, Grupp IL, Ji Y, Reed T, Loukianov E, Grupp G, Bhagwhat A, Hoit B, Walsh R, Marban E, Periasamy M. Targeted overexpression of the sarcoplasmic reticulum Ca2+-ATPase increases cardiac contractility in transgenic mouse hearts. Circulation Research. 1998;83:1205–1214. - PubMed
-
- Bers DM. Ryanodine and the calcium content of cardiac SR assessed by caffeine and rapid cooling contractures. American Journal of Physiology. 1987;253:C408–415. - PubMed
-
- Bokkala S, el Daher SS, Kakkar VV, Wuytack F, Authi KS. Localization and identification of Ca2+ATPases in highly purified human platelet plasma and intracellular membranes. Evidence that the monoclonal antibody PL/IM 430 recognizes the SERCA 3 Ca2+ATPase in human platelets. Biochemical Journal. 1995;306:837–842. - PMC - PubMed
-
- Brandl CJ, Green NM, Korczak B, MacLennan DH. Two Ca2+-ATPase genes: homologies and mechanistic implications of deduced amino acid sequences. Cell. 1986;44:597–607. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

