The initiation of the swing phase in human infant stepping: importance of hip position and leg loading
- PMID: 11034628
- PMCID: PMC2270131
- DOI: 10.1111/j.1469-7793.2000.00389.x
The initiation of the swing phase in human infant stepping: importance of hip position and leg loading
Abstract
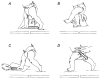
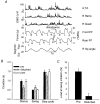
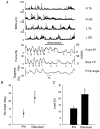
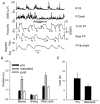
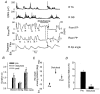
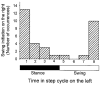
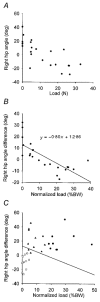
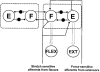
Hip extension and low load in the extensor muscles are important sensory signals that allow a decerebrate or spinal cat to advance from the stance phase to the swing phase during walking. We tested whether the same sensory information controlled the phases of stepping in human infants. Twenty-two infants between the ages of 5 and 12 months were studied during supported stepping on a treadmill. Forces exerted by the lower limbs, surface electromyography (EMG) from muscles, and the right hip angle were recorded. The whole experimental session was videotaped. The hip position and the amount of load experienced by the right limb were manipulated during stepping by changing the position of the foot during the stance phase or by applying manual pressure on the pelvic crest. Disturbances with different combinations of hip position and load were used. The stance phase was prolonged and the swing phase delayed when the hip was flexed and the load on the limb was high. In contrast, stance phase was shortened and swing advanced when the hip was extended and the load was low. The results were remarkably similar to those in reduced preparations of the cat. They thus suggest that the behaviour of the brainstem and spinal circuitry for walking may be similar between human infants and cats. There was an inverse relationship between hip position and load at the time of swing initiation, indicating the two factors combine to regulate the transition.
Figures


References
-
- Andersson O, Grillner S. Peripheral control of the cat’s step cycle. I. Phase dependent effects of ramp-movements of the hip during fictive locomotion. Acta Physiologica Scandinavica. 1981;113:89–101. - PubMed
-
- Andersson O, Grillner S. Peripheral control of the cat’s step cycle. II. Entrainment of the central pattern generators for locomotion by sinusoidal hip movements during fictive locomotion. Acta Physiologica Scandinavica. 1983;118:229–239. - PubMed
-
- Andersson O, Grillner S, Lindquist M, Zomlefer M. Peripheral control of the spinal pattern generators for locomotion in cat. Brain Research. 1978;150:625–630. - PubMed
-
- Bassler U. The femur-tibia control system of stick insects – a model system for the study of the neural basis of joint control. Brain Research Reviews. 1993;18:207–226. - PubMed
-
- Calancie B, Needham-Shropshire B, Jacobs P, Willer K, Zych G, Green BA. Involuntary stepping after chronic spinal cord injury. Evidence for a central pattern generator for locomotion in man. Brain. 1994;117:1143–1159. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

