Fumonisin B1-induced cell death in arabidopsis protoplasts requires jasmonate-, ethylene-, and salicylate-dependent signaling pathways
- PMID: 11041879
- PMCID: PMC149122
- DOI: 10.1105/tpc.12.10.1823
Fumonisin B1-induced cell death in arabidopsis protoplasts requires jasmonate-, ethylene-, and salicylate-dependent signaling pathways
Abstract
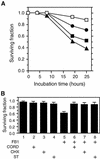
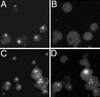
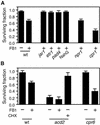
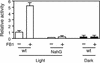
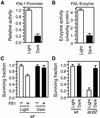
We have established an Arabidopsis protoplast model system to study plant cell death signaling. The fungal toxin fumonisin B1 (FB1) induces apoptosis-like programmed cell death (PCD) in wild-type protoplasts. FB1, however, only marginally affects the viability of protoplasts isolated from transgenic NahG plants, in which salicylic acid (SA) is metabolically degraded; from pad4-1 mutant plants, in which an SA amplification mechanism is thought to be impaired; or from jar1-1 or etr1-1 mutant plants, which are insensitive to jasmonate (JA) or ethylene (ET), respectively. FB1 susceptibility of wild-type protoplasts decreases in the dark, as does the cellular content of phenylalanine ammonia-lyase, a light-inducible enzyme involved in SA biosynthesis. Interestingly, however, FB1-induced PCD does not require the SA signal transmitter NPR1, given that npr1-1 protoplasts display wild-type FB1 susceptibility. Arabidopsis cpr1-1, cpr6-1, and acd2-2 protoplasts, in which the SA signaling pathway is constitutively activated, exhibit increased susceptibility to FB1. The cpr6-1 and acd2-2 mutants also constitutively express the JA and ET signaling pathways, but only the acd2-2 protoplasts undergo PCD in the absence of FB1. These results demonstrate that FB1 killing of Arabidopsis is light dependent and requires SA-, JA-, and ET-mediated signaling pathways as well as one or more unidentified factors activated by FB1 and the acd2-2 mutation.
Figures


References
-
- Bate, N.J., Orr, J., Ni, W., Meromi, A., Nadler-Hassar, T., Doerner, P.W., Dixon, R.A., Lamb, C.J., and Elkind, Y. (1994). Quantitative relationship between phenylalanine ammonia-lyase levels and phenylpropanoid accumulation in transgenic tobacco identifies a rate-determining step in natural product synthesis. Proc. Natl. Acad. Sci. USA 91, 7608–7612. - PMC - PubMed
-
- Bleecker, A.B., Estelle, M.A., Somerville, C., and Kende, H. (1988). Insensitivity to ethylene conferred by a dominant mutation in Arabidopsis thaliana. Science 241, 1086–1089. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

