Non-competitive pharmacological antagonism at the rabbit B(1) receptor
- PMID: 11053207
- PMCID: PMC1572413
- DOI: 10.1038/sj.bjp.0703656
Non-competitive pharmacological antagonism at the rabbit B(1) receptor
Abstract
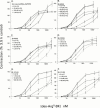
The B(1) receptor for kinins, stimulated by kinin metabolites without the C-terminal Arg residue (e.g., des-Arg(9)-bradykinin (BK) and Lys-des-Arg(9)-BK), is an increasingly recognized molecular target for the development of analgesic and anti-inflammatory drugs. Recently developed antagonists of this receptor were compared to a conventional antagonist, Ac-Lys-[Leu(8)]-des-Arg(9)-BK, in pharmacological assays based on the rabbit B(1) receptor. B-9858 (Lys-Lys-[Hyp(3), Igl(5), D-Igl(7), Oic(8)]des-Arg(9)-BK) and three other analogues possessing the alpha-2-indanylglycine(5) (Igl(5)) residue (order of potency B-9858 approximately B-10146>B-10148>B-10050) were partially insurmountable antagonists of des-Arg(9)-BK in the contractility assay based on rabbit aortic rings. B-9858-induced depression of the maximal effect was more pronounced in tissues treated with the protein synthesis inhibitor cycloheximide to block the spontaneous increase of response attributed to the post-isolation formation of B(1) receptors, and only partly reversible on washing. By comparison, Ac-Lys-[Leu(8)]des-Arg(9)-BK was a surmountable antagonist (pA(2) 7. 5), even in cycloheximide-treated tissues. B-9958 (Lys-[Hyp(3), CpG(5), D-Tic(7), CpG(8)]des-Arg(9)-BK) was also surmountable (pA(2) 8.5). The binding of [(3)H]-Lys-des-Arg(9)-BK to recombinant rabbit B(1) receptors expressed in COS-1 cells was influenced by two of the antagonists: while Ac-Lys-[Leu(8)]des-Arg(9)-BK competed for the radioligand binding without affecting the B(max), B-9858 decreased the B(max) in a time-dependent and washout-resistant manner. B-9858 and analogues possessing Igl(5) are the first reported non-competitive, non-equilibrium antagonists of the kinin B(1) receptor.
Figures




Similar articles
-
Endogenous aminopeptidase N decreases the potency of peptide agonists and antagonists of the kinin B1 receptors in the rabbit aorta.J Pharmacol Exp Ther. 2005 Sep;314(3):1169-76. doi: 10.1124/jpet.105.088799. Epub 2005 May 26. J Pharmacol Exp Ther. 2005. PMID: 15919764
-
Characterization of bradykinin receptors in a human osteoblastic cell line.Regul Pept. 2002 Jan 15;103(1):39-51. doi: 10.1016/s0167-0115(01)00325-1. Regul Pept. 2002. PMID: 11738247
-
Discovery of a dual-function peptide that combines aminopeptidase N inhibition and kinin B1 receptor antagonism.J Pharmacol Exp Ther. 2006 Apr;317(1):300-8. doi: 10.1124/jpet.105.095661. Epub 2005 Dec 20. J Pharmacol Exp Ther. 2006. PMID: 16368899
-
N-terminal extended conjugates of the agonists and antagonists of both bradykinin receptor subtypes: structure-activity relationship, cell imaging using ligands conjugated with fluorophores and prospect for functionally active cargoes.Peptides. 2012 Apr;34(2):433-46. doi: 10.1016/j.peptides.2012.02.007. Epub 2012 Feb 18. Peptides. 2012. PMID: 22349904 Review.
-
Potent, long-acting bradykinin antagonists for a wide range of applications.Can J Physiol Pharmacol. 1997 Jun;75(6):719-24. Can J Physiol Pharmacol. 1997. PMID: 9276154 Review.
Cited by
-
In Vivo Effects of Bradykinin B2 Receptor Agonists with Varying Susceptibility to Peptidases.Front Pharmacol. 2016 Jan 12;6:306. doi: 10.3389/fphar.2015.00306. eCollection 2015. Front Pharmacol. 2016. PMID: 26793104 Free PMC article.
-
Kinin B1 receptors: key G-protein-coupled receptors and their role in inflammatory and painful processes.Br J Pharmacol. 2004 Dec;143(7):803-18. doi: 10.1038/sj.bjp.0706012. Epub 2004 Nov 1. Br J Pharmacol. 2004. PMID: 15520046 Free PMC article. Review.
-
Absence of ligand-induced regulation of kinin receptor expression in the rabbit.Br J Pharmacol. 2001 Aug;133(7):1154-62. doi: 10.1038/sj.bjp.0704158. Br J Pharmacol. 2001. PMID: 11487527 Free PMC article.
References
-
- AHLUWALIA A., PERETTI M. B1 receptors as a new inflammatory target: Could this B the 1. Trends Pharmacol. Sci. 1999;20:100–104. - PubMed
-
- ARAMORI I., ZENKOH J., MORIKAWA N., O'DONNELL N., ASANO M., NAKAMURA K., IWAMI M., KOJO H., NOTSU Y. Novel subtype selective nonpeptide bradykinin receptor antagonists FR167344 and FR173657. Mol. Pharmacol. 1997;51:171–176. - PubMed
-
- AUDET R., PETITCLERC E., DRAPEAU G., RIOUX F., MARCEAU F. Further analysis of the upregulation of bradykinin B1 receptors in isolated rabbit aorta by using metabolic inhibitors. Eur. J. Pharmacol. 1994;271:551–555. - PubMed
-
- AUSTIN K.E., FAUSSNER A., ROBINSON H.E., CHAKRAVARTY S., KYLE D.J., BATHON J.M., PROUD D. Stable expression of the human kinin B1 receptor in Chinese hamster ovary cells. J. Biol. Chem. 1997;272:11420–11425. - PubMed
-
- BACHVAROV D.R., SAINT-JACQUES E., LARRIVÉE J.-F., LEVESQUE L., RIOUX F., DRAPEAU G., MARCEAU F. Cloning and pharmacological characterization of the rabbit bradykinin B2 receptor. J. Pharmacol. Exp. Ther. 1995;275:1623–1630. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

