Purification and cloning of cytotoxic ribonucleases from Rana catesbeiana (bullfrog)
- PMID: 11058105
- PMCID: PMC113159
- DOI: 10.1093/nar/28.21.4097
Purification and cloning of cytotoxic ribonucleases from Rana catesbeiana (bullfrog)
Abstract
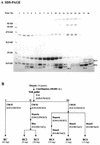
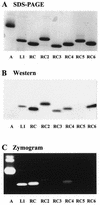
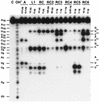
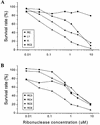
Ribonucleases with antitumor activity are mainly found in the oocytes and embryos of frogs, but the role of these ribonucleases in frog development is not clear. Moreover, most frog ribonuclease genes have not been cloned and characterized. In the present study, a group of ribonucleases were isolated from Rana catesbeiana (bullfrog). These ribonucleases in mature oocytes, namely RC-RNase, RC-RNase 2, RC-RNase 3, RC-RNase 4, RC-RNase 5 and RC-RNase 6, as well as liver-specific ribonuclease RC-RNase L1, were purified by column chromatographs and detected by zymogram assay and western blotting. Characterization of these purified ribonucleases revealed that they were highly conserved in amino acid sequence and had a pyroglutamate residue at their N-termini, but possessed different specific activities, base specificities and optimal pH values for their activities. These ribonucleases were cytotoxic to cervical carcinoma HeLa cells, but their cytotoxicities were not closely correlated to their enzymatic specific activities. Some other amino acid residues in addition to their catalytic residues were implicated to be involved in the cytotoxicity of the frog ribonucleases to tumor cells. Because the coding regions lack introns, the ribonuclease genes were cloned by PCR using genomic DNA as template. Their DNA sequences and amino acid sequences are homologous to those of mammalian ribonuclease superfamily, approximately 50 and approximately 25%, respectively.
Figures

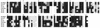
Similar articles
-
Large-scale preparation of a ribonuclease from Rana catesbeiana (bullfrog) oocytes and characterization of its specific cytotoxic activity against tumor cells.Protein Expr Purif. 1996 Mar;7(2):194-202. doi: 10.1006/prep.1996.0027. Protein Expr Purif. 1996. PMID: 8812861
-
The Rana catesbeiana rcr gene encoding a cytotoxic ribonuclease. Tissue distribution, cloning, purification, cytotoxicity, and active residues for RNase activity.J Biol Chem. 1998 Mar 13;273(11):6395-401. doi: 10.1074/jbc.273.11.6395. J Biol Chem. 1998. PMID: 9497370
-
Residues involved in the catalysis, base specificity, and cytotoxicity of ribonuclease from Rana catesbeiana based upon mutagenesis and X-ray crystallography.J Biol Chem. 2003 Feb 28;278(9):7300-9. doi: 10.1074/jbc.M206701200. Epub 2002 Dec 23. J Biol Chem. 2003. PMID: 12499382
-
Onconase and amphinase, the antitumor ribonucleases from Rana pipiens oocytes.Curr Pharm Biotechnol. 2008 Jun;9(3):215-25. doi: 10.2174/138920108784567245. Curr Pharm Biotechnol. 2008. PMID: 18673287 Free PMC article. Review.
-
Biochemistry of frog ribonucleases.Cell Mol Life Sci. 1998 Aug;54(8):775-84. doi: 10.1007/s000180050206. Cell Mol Life Sci. 1998. PMID: 9760986 Free PMC article. Review.
Cited by
-
1H, 13C and 15N resonance assignments and secondary structure of the cytotoxic protein RNase 4 from bullfrog Rana catesbeiana oocytes.J Biomol NMR. 2001 May;20(1):93-4. doi: 10.1023/a:1011255913840. J Biomol NMR. 2001. PMID: 11430762 No abstract available.
-
Removal of N-terminal methionine from recombinant proteins by engineered E. coli methionine aminopeptidase.Protein Sci. 2004 Jul;13(7):1802-10. doi: 10.1110/ps.04679104. Protein Sci. 2004. PMID: 15215523 Free PMC article.
-
Ribonucleases as potential modalities in anticancer therapy.Eur J Pharmacol. 2009 Dec 25;625(1-3):181-9. doi: 10.1016/j.ejphar.2009.06.067. Epub 2009 Oct 14. Eur J Pharmacol. 2009. PMID: 19825371 Free PMC article. Review.
-
Rana catesbeiana ribonuclease induces cell apoptosis via the caspase-9/-3 signaling pathway in human glioblastoma DBTRG, GBM8901 and GBM8401 cell lines.Oncol Lett. 2015 Jun;9(6):2471-2476. doi: 10.3892/ol.2015.3117. Epub 2015 Apr 15. Oncol Lett. 2015. PMID: 26137092 Free PMC article.
-
Sialic acid-binding lectin (leczyme) induces apoptosis to malignant mesothelioma and exerts synergistic antitumor effects with TRAIL.Int J Oncol. 2014 Feb;44(2):377-84. doi: 10.3892/ijo.2013.2192. Epub 2013 Nov 28. Int J Oncol. 2014. PMID: 24297392 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

