Age-associated characteristics of murine hematopoietic stem cells
- PMID: 11067876
- PMCID: PMC2193349
- DOI: 10.1084/jem.192.9.1273
Age-associated characteristics of murine hematopoietic stem cells
Abstract
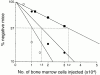
Little is known of age-associated functional changes in hematopoietic stem cells (HSCs). We studied aging HSCs at the clonal level by isolating CD34(-/low)c-Kit(+)Sca-1(+) lineage marker-negative (CD34(-)KSL) cells from the bone marrow of C57BL/6 mice. A population of CD34(-)KSL cells gradually expanded as age increased. Regardless of age, these cells formed in vitro colonies with stem cell factor and interleukin (IL)-3 but not with IL-3 alone. They did not form day 12 colony-forming unit (CFU)-S, indicating that they are primitive cells with myeloid differentiation potential. An in vivo limiting dilution assay revealed that numbers of multilineage repopulating cells increased twofold from 2 to 18 mo of age within a population of CD34(-)KSL cells as well as among unseparated bone marrow cells. In addition, we detected another compartment of repopulating cells, which differed from HSCs, among CD34(-)KSL cells of 18-mo-old mice. These repopulating cells showed less differentiation potential toward lymphoid cells but retained self-renewal potential, as suggested by secondary transplantation. We propose that HSCs gradually accumulate with age, accompanied by cells with less lymphoid differentiation potential, as a result of repeated self-renewal of HSCs.
Figures


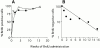
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

