Characterization of fission yeast cohesin: essential anaphase proteolysis of Rad21 phosphorylated in the S phase
- PMID: 11069892
- PMCID: PMC317025
- DOI: 10.1101/gad.832000
Characterization of fission yeast cohesin: essential anaphase proteolysis of Rad21 phosphorylated in the S phase
Abstract
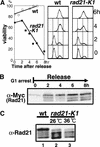
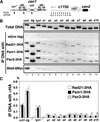
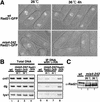
Cohesin complex acts in the formation and maintenance of sister chromatid cohesion during and after S phase. Budding yeast Scc1p/Mcd1p, an essential subunit, is cleaved and dissociates from chromosomes in anaphase, leading to sister chromatid separation. Most cohesin in higher eukaryotes, in contrast, is dissociated from chromosomes well before anaphase. The universal role of cohesin during anaphase thus remains to be determined. We report here initial characterization of four putative cohesin subunits, Psm1, Psm3, Rad21, and Psc3, in fission yeast. They are essential for sister chromatid cohesion. Immunoprecipitation demonstrates stable complex formation of Rad21 with Psm1 and Psm3 but not with Psc3. Chromatin immunoprecipitation shows that cohesin subunits are enriched in broad centromere regions and that the level of centromere-associated Rad21 did not change from metaphase to anaphase, very different from budding yeast. In contrast, Rad21 containing similar cleavage sites to those of Scc1p/Mcd1p is cleaved specifically in anaphase. This cleavage is essential, although the amount of cleaved product is very small (<5%). Mis4, another sister chromatid cohesion protein, plays an essential role for loading Rad21 on chromatin. A simple model is presented to explain the specific behavior of fission yeast cohesin and why only a tiny fraction of Rad21 is sufficient to be cleaved for normal anaphase.
Figures




References
-
- Birkenbihl RP, Subramani S. The rad21 gene product of Schizosaccharomyces pombe is a nuclear, cell cycle-regulated phosphoprotein. J Biol Chem. 1995;270:7703–7711. - PubMed
-
- Castano IB, Brzoska PM, Sadoff BU, Chen H, Christman MF. Mitotic chromosome condensation in the rDNA requires TRF4 and DNA topoisomerase I in Saccharomyces cerevisiae. Genes & Dev. 1996;10:2564–2576. - PubMed
-
- Ciosk R, Zachariae W, Michaelis C, Shevchenko A, Mann M, Nasmyth K. An ESP1/PDS1 complex regulates loss of sister chromatid cohesion at the metaphase to anaphase transition in yeast. Cell. 1998;93:1067–1076. - PubMed
-
- Ciosk R, Shirayama M, Shevchenko A, Tanaka T, Toth A, Shevchenko A, Nasmyth K. Cohesin's binding to chromosomes depends on a separate complex consisting of Scc2 and Scc4 proteins. Mol Cell. 2000;5:243–254. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
