Cloning and functional analysis of sucrose:sucrose 1-fructosyltransferase from tall fescue
- PMID: 11080298
- PMCID: PMC59220
- DOI: 10.1104/pp.124.3.1217
Cloning and functional analysis of sucrose:sucrose 1-fructosyltransferase from tall fescue
Abstract
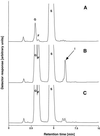
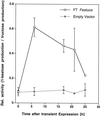
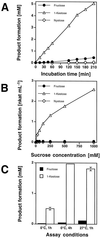
Enzymes of grasses involved in fructan synthesis are of interest since they play a major role in assimilate partitioning and allocation, for instance in the leaf growth zone. Several fructosyltransferases from tall fescue (Festuca arundinacea) have previously been purified (Lüscher and Nelson, 1995). It is surprising that all of these enzyme preparations appeared to act both as sucrose (Suc):Suc 1-fructosyl transferases (1-SST) and as fructan:fructan 6(G)-fructosyl transferases. Here we report the cloning of a cDNA corresponding to the predominant protein in one of the fructosyl transferase preparations, its transient expression in tobacco protoplasts, and its functional analysis in the methylotrophic yeast, Pichia pastoris. When the cDNA was transiently expressed in tobacco protoplasts, the corresponding enzyme preparations produced 1-kestose from Suc, showing that the cDNA encodes a 1-SST. When the cDNA was expressed in P. pastoris, the recombinant protein had all the properties of known 1-SSTs, namely 1-kestose production, moderate nystose production, lack of 6-kestose production, and fructan exohydrolase activity with 1-kestose as the substrate. The physical properties were similar to those of the previously purified enzyme, except for its apparent lack of fructan:fructan 6(G)-fructosyl transferase activity. The expression pattern of the corresponding mRNA was studied in different zones of the growing leaves, and it was shown that transcript levels matched the 1-SST activity and fructan content.
Figures




Similar articles
-
Cloning of the fructan biosynthesis pathway of Jerusalem artichoke.Plant J. 1998 Aug;15(4):489-500. doi: 10.1046/j.1365-313x.1998.00230.x. Plant J. 1998. PMID: 9753774
-
Cloning of sucrose:sucrose 1-fructosyltransferase from onion and synthesis of structurally defined fructan molecules from sucrose.Plant Physiol. 1998 Aug;117(4):1507-13. doi: 10.1104/pp.117.4.1507. Plant Physiol. 1998. PMID: 9701606 Free PMC article.
-
Unexpected presence of graminan- and levan-type fructans in the evergreen frost-hardy eudicot Pachysandra terminalis (Buxaceae): purification, cloning, and functional analysis of a 6-SST/6-SFT enzyme.Plant Physiol. 2011 Jan;155(1):603-14. doi: 10.1104/pp.110.162222. Epub 2010 Oct 29. Plant Physiol. 2011. PMID: 21037113 Free PMC article.
-
Interaction between fructan metabolism and plant growth regulators.Planta. 2022 Jan 27;255(2):49. doi: 10.1007/s00425-022-03826-1. Planta. 2022. PMID: 35084581 Review.
-
Engineering fructan metabolism in plants.J Plant Physiol. 2003 Jul;160(7):811-20. doi: 10.1078/0176-1617-01029. J Plant Physiol. 2003. PMID: 12940548 Review.
Cited by
-
Ectopic Expression of the Allium cepa 1-SST Gene in Cotton Improves Drought Tolerance and Yield Under Drought Stress in the Field.Front Plant Sci. 2022 Jan 12;12:783134. doi: 10.3389/fpls.2021.783134. eCollection 2021. Front Plant Sci. 2022. PMID: 35095957 Free PMC article.
-
Towards a better understanding of the generation of fructan structure diversity in plants: molecular and functional characterization of a sucrose:fructan 6-fructosyltransferase (6-SFT) cDNA from perennial ryegrass (Lolium perenne).J Exp Bot. 2011 Mar;62(6):1871-85. doi: 10.1093/jxb/erq388. Epub 2010 Dec 31. J Exp Bot. 2011. PMID: 21196473 Free PMC article.
-
Effects of different carbohydrate sources on fructan metabolism in plants of Chrysolaena obovata grown in vitro.Front Plant Sci. 2015 Sep 7;6:681. doi: 10.3389/fpls.2015.00681. eCollection 2015. Front Plant Sci. 2015. PMID: 26442003 Free PMC article.
-
Fructans redistribution prior to sprouting in stored onion bulbs is a potential marker for dormancy break.Postharvest Biol Technol. 2019 Mar;149:221-234. doi: 10.1016/j.postharvbio.2018.12.002. Postharvest Biol Technol. 2019. PMID: 30828134 Free PMC article.
-
Fructan and its relationship to abiotic stress tolerance in plants.Cell Mol Life Sci. 2009 Jul;66(13):2007-23. doi: 10.1007/s00018-009-0002-x. Epub 2009 Mar 17. Cell Mol Life Sci. 2009. PMID: 19290476 Free PMC article. Review.
References
-
- Clare JJ, Romanos MA, Rayment FB, Rowedder JE, Smith MA, Payne MM, Sreekrishna K, Henwood CA. Production of mouse epidermal growth factor in yeast: high-level secretion using Pichia pastoris strains containing multiple gene copies. Gene. 1991;105:205–212. - PubMed
-
- Edelman J, Jefford TG. The mechanism of fructosan metabolism in higher plants as exemplified in Helianthus tuberosus L. New Phytol. 1968;67:517–531.
-
- Goodall G, Kiss T, W. F. Nuclear RNA splicing and small nuclear RNAs and their genes in higher plants. Oxf Surv Plant Mol Cell Biol. 1991;7:255–296.
-
- Hawker JS, Jenner CF, Niemietz CM. Sugar metabolism and compartmentation. Aust J Plant Physiol. 1991;18:227–237.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

