Chitinase genes responsive to cold encode antifreeze proteins in winter cereals
- PMID: 11080301
- PMCID: PMC59223
- DOI: 10.1104/pp.124.3.1251
Chitinase genes responsive to cold encode antifreeze proteins in winter cereals
Abstract
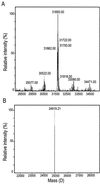
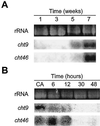
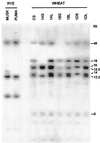
Antifreeze proteins similar to two different chitinases accumulate during cold acclimation in winter rye (Secale cereale). To determine whether these cold-responsive chitinases require post-translational modification to bind to ice, cDNAs coding for two different full-length chitinases were isolated from a cDNA library produced from cold-acclimated winter rye leaves. CHT9 is a 1,193-bp clone that encodes a 31.7-kD class I chitinase and CHT46 is a 998-bp clone that codes for a 24.8-kD class II chitinase. Chitinase-antifreeze proteins purified from the plant were similar in mass to the predicted mature products of CHT9 and CHT46, thus indicating that there was little chemical modification of the amino acid sequences in planta. To confirm these results, the mature sequences of CHT9 and CHT46 were expressed in Escherichia coli and the products of both cDNAs modified the growth of ice. Transcripts of both genes accumulated late in cold acclimation in winter rye. Southern analysis of winter rye genomic DNA indicated the presence of a small gene family homologous to CHT46. In hexaploid wheat, CHT46 homologs mapped to the homeologous group 1 chromosomes and were expressed in response to cold and drought. We conclude that two novel cold-responsive genes encoding chitinases with ice-binding activity may have arisen in winter rye and other cereals through gene duplication.
Figures





References
-
- Altschul SF, Gish W, Miller W, Meyers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215:403–410. - PubMed
-
- Antikainen M, Griffith M. Antifreeze protein accumulation in freezing-tolerant cereals. Physiol Plant. 1997;99:423–432.
-
- Beintema JJ. Structural feature of plant chitinases and chitin-binding proteins. FEBS Lett. 1994;350:159–163. - PubMed
-
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:341–374. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

