Inward potassium channel in guard cells as a target for polyamine regulation of stomatal movements
- PMID: 11080307
- PMCID: PMC59229
- DOI: 10.1104/pp.124.3.1315
Inward potassium channel in guard cells as a target for polyamine regulation of stomatal movements
Abstract
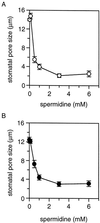
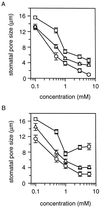
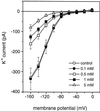
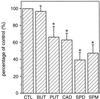
A number of studies show that environmental stress conditions such as drought, high salt, and air pollutants increase polyamine levels in plant cells. However, little is understood about the physiological function of elevated polyamine levels. We report here that polyamines regulate the voltage-dependent inward K(+) channel in the plasma membrane of guard cells and modulate stomatal aperture, a plant "sensor" to environmental changes. All natural polyamines, including spermidine, spermine, cadaverine, and putrescine, strongly inhibited opening and induced closure of stomata. Whole-cell patch-clamp analysis showed that intracellular application of polyamines inhibited the inward K(+) current across the plasma membrane of guard cells. Single-channel recording analysis indicated that polyamine regulation of the K(+) channel requires unknown cytoplasmic factors. In an effort to identify the target channel at the molecular level, we found that spermidine inhibited the inward K(+) current carried by KAT1 channel that was functionally expressed in a plant cell model. These findings suggest that polyamines target KAT1-like inward K(+) channels in guard cells and modulate stomatal movements, providing a link between stress conditions, polyamine levels, and stomatal regulation.
Figures



Similar articles
-
The mechanism of inward rectification of potassium channels: "long-pore plugging" by cytoplasmic polyamines.J Gen Physiol. 1995 Nov;106(5):923-55. doi: 10.1085/jgp.106.5.923. J Gen Physiol. 1995. PMID: 8648298 Free PMC article.
-
Distinct abscisic acid signaling pathways for modulation of guard cell versus mesophyll cell potassium channels revealed by expression studies in Xenopus laevis oocytes.Plant Physiol. 2000 Sep;124(1):223-30. doi: 10.1104/pp.124.1.223. Plant Physiol. 2000. PMID: 10982437 Free PMC article.
-
Dominant negative guard cell K+ channel mutants reduce inward-rectifying K+ currents and light-induced stomatal opening in arabidopsis.Plant Physiol. 2001 Oct;127(2):473-85. Plant Physiol. 2001. PMID: 11598222 Free PMC article.
-
Structure and function of potassium channels in plants: some inferences about the molecular origin of inward rectification in KAT1 channels (Review).Mol Membr Biol. 2003 Jan-Mar;20(1):19-25. doi: 10.1080/0968768021000057371. Mol Membr Biol. 2003. PMID: 12745922 Review.
-
Interactions of polyamines with ion channels.Biochem J. 1997 Jul 15;325 ( Pt 2)(Pt 2):289-97. doi: 10.1042/bj3250289. Biochem J. 1997. PMID: 9230104 Free PMC article. Review.
Cited by
-
The guard cell metabolome: functions in stomatal movement and global food security.Front Plant Sci. 2015 May 19;6:334. doi: 10.3389/fpls.2015.00334. eCollection 2015. Front Plant Sci. 2015. PMID: 26042131 Free PMC article. Review.
-
Polyamine Metabolism in Scots Pine Embryogenic Cells under Potassium Deficiency.Cells. 2021 May 18;10(5):1244. doi: 10.3390/cells10051244. Cells. 2021. PMID: 34070116 Free PMC article.
-
Spermine mediated improvements on stomatal features, growth, grain filling and yield of rice under differing water availability.Sci Rep. 2021 May 21;11(1):10669. doi: 10.1038/s41598-021-89812-1. Sci Rep. 2021. PMID: 34021188 Free PMC article.
-
Arabidopsis thickvein mutation affects vein thickness and organ vascularization, and resides in a provascular cell-specific spermine synthase involved in vein definition and in polar auxin transport.Plant Physiol. 2005 Jun;138(2):767-77. doi: 10.1104/pp.104.055756. Epub 2005 May 13. Plant Physiol. 2005. PMID: 15894745 Free PMC article.
-
Exogenous putrescine attenuates the negative impact of drought stress by modulating physio-biochemical traits and gene expression in sugar beet (Beta vulgaris L.).PLoS One. 2022 Jan 7;17(1):e0262099. doi: 10.1371/journal.pone.0262099. eCollection 2022. PLoS One. 2022. PMID: 34995297 Free PMC article.
References
-
- Assmann SM. Signal transduction in guard cells. Annu Rev Cell Biol. 1993;9:345–375. - PubMed
-
- Bei QX, Luan S. Functional expression and characterization of an inward K+ channel gene in a plant cell model. Plant J. 1998;13:857–865. - PubMed
-
- Blatt MR. Ion channel gating in plants: physiological implications and integration for stomatal function. J Membr Biol. 1991;124:95–112. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

