The role of vacuolar malate-transport capacity in crassulacean acid metabolism and nitrate nutrition. Higher malate-transport capacity in ice plant after crassulacean acid metabolism-induction and in tobacco under nitrate nutrition
- PMID: 11080309
- PMCID: PMC59231
- DOI: 10.1104/pp.124.3.1335
The role of vacuolar malate-transport capacity in crassulacean acid metabolism and nitrate nutrition. Higher malate-transport capacity in ice plant after crassulacean acid metabolism-induction and in tobacco under nitrate nutrition
Abstract
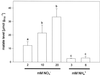
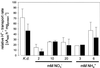
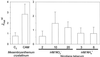
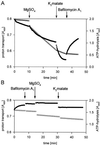
Anion uptake by isolated tonoplast vesicles was recorded indirectly via increased H(+)-transport by H(+)-pumping of the V-ATPase due to dissipation of the electrical component of the electrochemical proton gradient, Deltamu(H+), across the membrane. ATP hydrolysis by the V-ATPase was measured simultaneously after the Palmgren test. Normalizing for ATP-hydrolysis and effects of chloride, which was added to the assays as a stimulating effector of the V-ATPase, a parameter, J(mal)(rel), of apparent ATP-dependent malate-stimulated H(+)-transport was worked out as an indirect measure of malate transport capacity. This allowed comparison of various species and physiological conditions. J(mal)(rel) was high in the obligate crassulacean acid metabolism (CAM) species Kalanchoë daigremontiana Hamet et Perrier, it increased substantially after CAM induction in ice plant (Mesembryanthemum crystallinum), and it was positively correlated with NO(3)(-) nutrition in tobacco (Nicotiana tabacum). For tobacco this was confirmed by measurements of malate transport energized via the V-PPase. In ice plant a new polypeptide of 32-kD apparent molecular mass appeared, and a 33-kD polypeptide showed higher levels after CAM induction under conditions of higher J(mal)(rel). It is concluded that tonoplast malate transport capacity plays an important role in physiological regulation in CAM and NO(3)(-) nutrition and that a putative malate transporter must be within the 32- to 33-kD polypeptide fraction of tonoplast proteins.
Figures






Similar articles
-
The 32 kDa tonoplast polypeptide Di associated with the V-type H+-ATPase of Mesembryanthemum crystallinum L. in the CAM state: A proteolytically processed subunit B?FEBS Lett. 1996 Jul 8;389(3):314-8. doi: 10.1016/0014-5793(96)00556-x. FEBS Lett. 1996. PMID: 8766723
-
Changes in acidity and in proton transport at the tonoplast of grape berries during development.Planta. 2001 May;213(1):20-8. doi: 10.1007/s004250000472. Planta. 2001. PMID: 11523652
-
Differential immunological cross-reactions with antisera against the V-ATPase of Kalanchoë daigremontiana reveal structural differences of V-ATPase subunits of different plant species.Biol Chem. 1997 Oct;378(10):1131-9. doi: 10.1515/bchm.1997.378.10.1131. Biol Chem. 1997. PMID: 9372182
-
Vacuolar and plasma membrane proton-adenosinetriphosphatases.Physiol Rev. 1999 Apr;79(2):361-85. doi: 10.1152/physrev.1999.79.2.361. Physiol Rev. 1999. PMID: 10221984 Review.
-
Plant proton pumps.FEBS Lett. 2007 May 25;581(12):2204-14. doi: 10.1016/j.febslet.2007.03.050. Epub 2007 Mar 30. FEBS Lett. 2007. PMID: 17412324 Review.
Cited by
-
NO₃⁻/H⁺ antiport in the tonoplast of cucumber root cells is stimulated by nitrate supply: evidence for a reversible nitrate-induced phosphorylation of vacuolar NO₃⁻/H⁺ antiport.PLoS One. 2013 Sep 11;8(9):e73972. doi: 10.1371/journal.pone.0073972. eCollection 2013. PLoS One. 2013. PMID: 24040130 Free PMC article.
-
Na+/H+-transporter, H+-pumps and an aquaporin in light and heavy tonoplast membranes from organic acid and NaCl accumulating vacuoles of the annual facultative CAM plant and halophyte Mesembryanthemum crystallinum L.Planta. 2006 Sep;224(4):944-51. doi: 10.1007/s00425-006-0265-5. Epub 2006 Mar 31. Planta. 2006. PMID: 16575596
-
Plant nitrogen uptake and assimilation: regulation of cellular pH homeostasis.J Exp Bot. 2020 Jul 25;71(15):4380-4392. doi: 10.1093/jxb/eraa150. J Exp Bot. 2020. PMID: 32206788 Free PMC article. Review.
-
Machine Learning Unmasked Nutritional Imbalances on the Medicinal Plant Bryophyllum sp. Cultured in vitro.Front Plant Sci. 2020 Dec 1;11:576177. doi: 10.3389/fpls.2020.576177. eCollection 2020. Front Plant Sci. 2020. PMID: 33329638 Free PMC article.
-
Ammonium intensifies CAM photosynthesis and counteracts drought effects by increasing malate transport and antioxidant capacity in Guzmania monostachia.J Exp Bot. 2018 Apr 9;69(8):1993-2003. doi: 10.1093/jxb/ery054. J Exp Bot. 2018. PMID: 29462338 Free PMC article.
References
-
- Allen S, Raven JA. Intracellular pH regulation in Ricinus communis grown with ammonium or nitrate as N source: the role of long distance transport. J Exp Bot. 1987;38:580–596.
-
- Ames B. Assay of inorganic phosphate, total phosphate and phosphatase. Methods Enzymol. 1966;8:115–118.
-
- Bennett A, Spanswick R. Optical measurement of ΔpH and ΔΨ in corn root membrane vesicles: kinetic analysis of Cl− effects on proton translocating ATPase. J Membr Biol. 1983;71:95–107.
-
- Blake MS, Johnston KH, Russell-Jones GJ, Gotschlich EC. A rapid, sensitive method for detection of alkaline phosphatase-conjugated anti-antibody on western blots. Anal Biochem. 1984;136:175–179. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

