Identification of discrete domains within gonococcal transferrin-binding protein A that are necessary for ligand binding and iron uptake functions
- PMID: 11083823
- PMCID: PMC97808
- DOI: 10.1128/IAI.68.12.6988-6996.2000
Identification of discrete domains within gonococcal transferrin-binding protein A that are necessary for ligand binding and iron uptake functions
Abstract
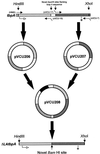
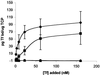
The availability of free iron in vivo is strictly limited, in part by the iron-binding protein transferrin. The pathogenic Neisseria spp. can sequester iron from this protein, dependent upon two iron-repressible, transferrin-binding proteins (TbpA and TbpB). TbpA is a TonB-dependent, integral, outer membrane protein that may form a beta-barrel exposing multiple surface loops, some of which are likely to contain ligand-binding motifs. In this study we propose a topological model of gonococcal TbpA and then test some of the hypotheses set forth by the model by individually deleting three putative loops (designated loops 4, 5, and 8). Each mutant TbpA could be expressed without toxicity and was surface exposed as assessed by immunoblotting, transferrin binding, and protease accessibility. Deletion of loop 4 or loop 5 abolished transferrin binding to whole cells in solid- and liquid-phase assays, while deletion of loop 8 decreased the affinity of the receptor for transferrin without affecting the copy number. Strains expressing any of the three mutated TbpAs were incapable of growth on transferrin as a sole iron source. These data implicate putative loops 4 and 5 as critical determinants for receptor function and transferrin-iron uptake by gonococcal TbpA. The phenotype of the DeltaL8TbpA mutant suggests that high-affinity ligand interaction is required for transferrin-iron internalization.
Figures




References
-
- Biswas G D, Anderson J E, Sparling P F. Cloning and functional characterization of Neisseria gonorrhoeae tonB, exbB, and exbD genes. Mol Microbiol. 1997;24:169–179. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

