Anopheles gambiae salivary gland proteins as putative targets for blocking transmission of malaria parasites
- PMID: 11087838
- PMCID: PMC17666
- DOI: 10.1073/pnas.250472597
Anopheles gambiae salivary gland proteins as putative targets for blocking transmission of malaria parasites
Abstract
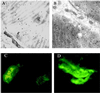
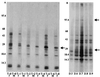
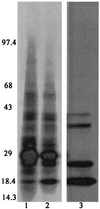
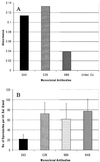
Anopheles gambiae is the primary vector of human malaria in sub-Saharan Africa. Invasion of Anopheles salivary glands by Plasmodium sporozoites is a necessary step in the transmission of malaria and is likely to be mediated by specific receptor-ligand interactions. We are interested in identifying putative an A. gambiae salivary gland receptor or receptors for sporozoite invasion as a possible target for blocking malaria transmission. By using monoclonal antibodies against female-specific A. gambiae salivary gland proteins, two molecules, one of 29 kDa and one of 100 kDa, were identified and characterized with respect to the age and blood-feeding process of mosquitoes. In an in vivo bioassay, the monoclonal antibody against the 100-kDa protein inhibited Plasmodium yoelii sporozoite invasion of salivary glands >/=75%. These results show that A. gambiae salivary gland proteins are accessible to monoclonal antibodies that inhibit sporozoite invasion of the salivary glands and suggest alternate targets for blocking the transmission of malaria by this most competent of malaria vectors.
Figures

Similar articles
-
Total and putative surface proteomics of malaria parasite salivary gland sporozoites.Mol Cell Proteomics. 2013 May;12(5):1127-43. doi: 10.1074/mcp.M112.024505. Epub 2013 Jan 16. Mol Cell Proteomics. 2013. PMID: 23325771 Free PMC article.
-
Plasmodium gallinaceum: sporozoite invasion of Aedes aegypti salivary glands is inhibited by anti-gland antibodies and by lectins.Exp Parasitol. 1995 Nov;81(3):332-43. doi: 10.1006/expr.1995.1124. Exp Parasitol. 1995. PMID: 7498430
-
Plasmodium vivax: a monoclonal antibody recognizes a circumsporozoite protein precursor on the sporozoite surface.Exp Parasitol. 1998 Nov;90(3):203-11. doi: 10.1006/expr.1998.4334. Exp Parasitol. 1998. PMID: 9806864
-
Salivary gland-specific gene expression in the malaria vector Anopheles gambiae.Parassitologia. 1999 Sep;41(1-3):483-7. Parassitologia. 1999. PMID: 10697906 Review.
-
Malaria parasite development in mosquitoes.Annu Rev Entomol. 1998;43:519-43. doi: 10.1146/annurev.ento.43.1.519. Annu Rev Entomol. 1998. PMID: 9444756 Review.
Cited by
-
When Is a Plasmodium-Infected Mosquito an Infectious Mosquito?Trends Parasitol. 2020 Aug;36(8):705-716. doi: 10.1016/j.pt.2020.05.011. Epub 2020 Jul 1. Trends Parasitol. 2020. PMID: 32620501 Free PMC article. Review.
-
Analysis of female salivary gland proteins of the Anopheles barbirostris complex (Diptera: Culicidae) in Thailand.Parasitol Res. 2010 Aug;107(3):509-16. doi: 10.1007/s00436-010-1883-1. Epub 2010 Apr 29. Parasitol Res. 2010. PMID: 20428888
-
Dengue viruses binding proteins from Aedes aegypti and Aedes polynesiensis salivary glands.Virol J. 2009 Mar 25;6:35. doi: 10.1186/1743-422X-6-35. Virol J. 2009. PMID: 19320997 Free PMC article.
-
Defining the genetics of the widely used G3 strain of the mosquito, Anopheles gambiae.Sci Rep. 2025 Apr 16;15(1):13142. doi: 10.1038/s41598-025-96391-y. Sci Rep. 2025. PMID: 40240469 Free PMC article.
-
The salivary protein Saglin facilitates efficient midgut colonization of Anopheles mosquitoes by malaria parasites.PLoS Pathog. 2023 Mar 2;19(3):e1010538. doi: 10.1371/journal.ppat.1010538. eCollection 2023 Mar. PLoS Pathog. 2023. PMID: 36862755 Free PMC article.
References
-
- Rosenberg R. Am J Trop Med Hyg. 1985;34:687–691. - PubMed
-
- Perrone J B, DeMaio J, Spielman A. Insect Biochem Mol Biol. 1986;16:313–318.
-
- Barreau C, Touray M, Pimenta P F, Miller L H, Vernick K D. Exp Parasitol. 1995;81:332–343. - PubMed
-
- Service M W. In: Bruce-Chwatt's Essential Malariology. Gilles H M, Warrell D A, editors. London: Edward Arnold; 1993.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

