Remodeling of alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptor subunit composition in hippocampal neurons after global ischemia
- PMID: 11087875
- PMCID: PMC27229
- DOI: 10.1073/pnas.97.24.13360
Remodeling of alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptor subunit composition in hippocampal neurons after global ischemia
Erratum in
- Proc Natl Acad Sci U S A 2001 Jan 30;98(3):1317. Optiz T [corrected to Opitz T]
Abstract
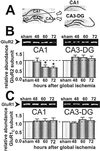
Transient global ischemia induces selective delayed cell death, primarily of principal neurons in the hippocampal CA1. However, the molecular mechanisms underlying ischemia-induced cell death are as yet unclear. The present study shows that global ischemia triggers a pronounced and cell-specific reduction in GluR2 [the subunit that limits Ca(2+) permeability of alpha-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) receptors] in vulnerable CA1 neurons, as evidenced by immunofluorescence of brain sections and Western blot analysis of microdissected hippocampal subfields. At 72 h after ischemia (a time before cell death), virtually all CA1 pyramidal neurons exhibited greatly reduced GluR2 immunolabeling throughout their somata and dendritic processes. GluR2 immunolabeling was unchanged in pyramidal cells of the CA3 and granule cells of the dentate gyrus, regions resistant to ischemia-induced damage. Immunolabeling of the AMPA receptor subunit GluR1 was unchanged in CA1, CA3, and dentate gyrus. Western analysis indicated that GluR2 subunit abundance was markedly reduced in CA1 at 60 and 72 h after the ischemic insult; GluR1 abundance was unchanged in all subfields at all times examined. These findings, together with the previous observation of enhanced AMPA-elicited Ca(2+) influx in postischemic CA1 neurons, show that functional GluR2-lacking, Ca(2+)-permeable AMPA receptors are expressed in vulnerable neurons before cell death. Thus, the present study provides an important link in the postulated causal chain between global ischemia and delayed death of CA1 pyramidal neurons.
Figures


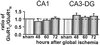
References
-
- Choi D W. Trends Neurosci. 1995;18:58–60. - PubMed
-
- Pulsinelli W A, Brierley J B, Plum F. Ann Neurol. 1982;11:491–498. - PubMed
-
- Seeburg P H. Trends Neurosci. 1993;16:359–365. - PubMed
-
- Verdoorn T A, Burnashev N, Monyer H, Seeburg P H, Sakmann B. Science. 1991;252:1715–1718. - PubMed
-
- Geiger J R, Melcher T, Koh D S, Sakmann B, Seeburg P H, Jonas P, Monyer H. Neuron. 1995;15:193–204. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

