[Nphe(1)]-Nociceptin (1-13)-NH(2), a nociceptin receptor antagonist, reverses nociceptin-induced spatial memory impairments in the Morris water maze task in rats
- PMID: 11090110
- PMCID: PMC1572479
- DOI: 10.1038/sj.bjp.0703724
[Nphe(1)]-Nociceptin (1-13)-NH(2), a nociceptin receptor antagonist, reverses nociceptin-induced spatial memory impairments in the Morris water maze task in rats
Abstract
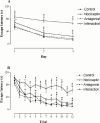
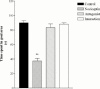
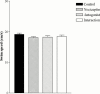
1. The present study was undertaken to investigate the effects of the novel nociceptin receptor antagonist, [Nphe(1)]-Nociceptin (1-13)-NH(2) (bilateral intrahippocampal injection, 50 nmole rat(-1)) on purported nociceptin-induced (bilateral intrahippocampal injection, 5 nmole rat(-1)) deficits in spatial learning in the rat Morris water maze task. In addition, experiments were performed in an 'open field' to investigate possible peptide-induced changes in exploratory behaviour. 2. Nociceptin significantly impaired the ability of the animal to locate the hidden platform throughout training (P<0.001 versus control group). 3. Pretreatment with [Nphe(1)]-Nociceptin (1-13)-NH(2) significantly blocked nociceptin-induced impairment of spatial learning (P<0.001 versus nociceptin group). 4. A probe trial revealed that vehicle-treated animals spent more time in the quadrant that had previously contained the hidden platform, whereas nociceptin-treated animals did not spend more time in any one quadrant. 5. Learning impairments were not attributable to non-specific deficits in motor performance or change in exploratory behaviour. 6. Taken together, our results reveal that [Nphe(1)]-Nociceptin (1-13)-NH(2) represents an effective and useful in vivo antagonist at the nociceptin receptors involved in learning and memory.
Figures


References
-
- BUNZOW J.R., SAEZ C., MORTRUD M., BOUVIER C., WILLIAMS J.T., LOW M., GRANDY D.K. Molecular cloning and tissue distribution of a putitive member of the rat opioid receptor gene family that is not mu, delta or kappa opioid receptor type. FEBS Lett. 1994;347:284–288. - PubMed
-
- CHEN Y., FAN Y., LIU J., MESTEK A., TIAN M., KOZAK C.A., YU L. Molecular cloning, tissue distribution and chromosomal localization of a novel member of the opioid receptor gene family. FEBS Lett. 1994;347:279–283. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

