Enzyme mimicry by the antiidiotypic antibody approach
- PMID: 11095704
- PMCID: PMC17609
- DOI: 10.1073/pnas.200360497
Enzyme mimicry by the antiidiotypic antibody approach
Abstract
The concept of "internal image" of antiidiotypic antibodies has provided the basis for eliciting catalytic antibodies. A monoclonal IgM 9A8 that was obtained as an antiidiotype to AE-2 mAb, a known inhibitor of acetylcholinesterase, displayed esterolytic activity. Study of recombinant Fab fragments and separate light and heavy chains of 9A8 confirmed that the antibody variable domain encodes the catalytic function, whereas neither part of the primary sequence of the Fab exhibited homology with the enzyme. The specific modification of the 9A8 variable domain by an active site-directed covalent inhibitor revealed the presence of an active site Ser residue. A three-dimensional modeling suggests the existence of a functional catalytic dyad Ser-His. Comparison of active sites of 9A8 and 17E8 esterolytic abzyme raised against transition-state analog revealed structural similarity although both antibodies were elicited by two different approaches.
Figures


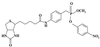




References
-
- Jencks W P. Catalysis in Chemistry and Enzymology. New York: McGraw–Hill; 1969. p. 288.
-
- Tramontano A, Janda K D, Lerner R A. Science. 1986;234:1566–1570. - PubMed
-
- Pollack S J, Jacobs J W, Schultz P G. Science. 1986;234:1570–1573. - PubMed
-
- Jerne N K. Ann Immunol. 1974;125C:373–389. - PubMed
-
- Pan Y, Yuhasz S C, Amzel L M. FASEB J. 1995;9:43–49. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources

