Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model
- PMID: 11095710
- PMCID: PMC17641
- DOI: 10.1073/pnas.240335297
Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model
Abstract
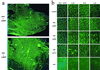
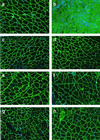
Duchenne muscular dystrophy (DMD) is the most common and lethal genetic muscle disorder, caused by recessive mutations in the dystrophin gene. One of every 3,500 males suffers from DMD, yet no treatment is currently available. Genetic therapeutic approaches, using primarily myoblast transplantation and adenovirus-mediated gene transfer, have met with limited success. Adeno-associated virus (AAV) vectors, although proven superior for muscle gene transfer, are too small (5 kb) to package the 14-kb dystrophin cDNA. Here we have created a series of minidystrophin genes (<4.2 kb) under the control of a muscle-specific promoter that readily package into AAV vectors. When injected into the muscle of mdx mice (a DMD model), two of the minigenes resulted in efficient and stable expression in a majority of the myofibers, restoring the missing dystrophin and dystrophin-associated protein complexes onto the plasma membrane. More importantly, this AAV treatment ameliorated dystrophic pathology in mdx muscle and led to normal myofiber morphology, histology, and cell membrane integrity. Thus, we have defined minimal functional dystrophin units and demonstrated the effectiveness of using AAV to deliver the minigenes in vivo, offering a promising avenue for DMD gene therapy.
Figures


Comment in
-
The dystrophin-associated glycoprotein complex: what parts can you do without?Proc Natl Acad Sci U S A. 2000 Dec 5;97(25):13464-6. doi: 10.1073/pnas.011510597. Proc Natl Acad Sci U S A. 2000. PMID: 11095702 Free PMC article. No abstract available.
References
-
- Kunkel L M. Nature (London) 1986;322:73–77. - PubMed
-
- Koenig M, Hoffman E P, Bertelson C J, Monaco A P, Feener C, Kunkel L M. Cell. 1987;50:509–517. - PubMed
-
- Hoffman E P, Brown R H, Jr, Kunkel L M. Cell. 1987;51:919–928. - PubMed
-
- Koenig M, Kunkel L M. J Biol Chem. 1990;265:4560–4566. - PubMed
-
- Gussoni E, Blau H M, Kunkel L M. Nat Med. 1997;3:970–977. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

