A novel assembly mechanism for the DNA polymerase III holoenzyme DnaX complex: association of deltadelta' with DnaX(4) forms DnaX(3)deltadelta'
- PMID: 11101526
- PMCID: PMC305859
- DOI: 10.1093/emboj/19.23.6536
A novel assembly mechanism for the DNA polymerase III holoenzyme DnaX complex: association of deltadelta' with DnaX(4) forms DnaX(3)deltadelta'
Abstract
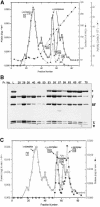
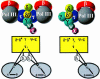
We have constructed a plasmid-borne artificial operon that expresses the six subunits of the DnaX complex of Escherichia coli DNA polymerase III holoenzyme: tau, gamma, delta, delta', chi and psi. Induction of this operon followed by assembly in vivo produced two taugamma mixed DnaX complexes with stoichiometries of tau(1)gamma(2)deltadelta'chipsi and tau(2)gamma(1)deltadelta'chipsi rather than the expected gamma(2)tau(2)deltadelta'chipsi. We observed the same heterogeneity when taugamma mixed DnaX complexes were reconstituted in vitro. Re-examination of homomeric DnaX tau and gamma complexes assembled either in vitro or in vivo also revealed a stoichiometry of DnaX(3)deltadelta'chipsi. Equilibrium sedimentation analysis showed that free DnaX is a tetramer in equilibrium with a free monomer. An assembly mechanism, in which the association of heterologous subunits with a homomeric complex alters the stoichiometry of the homomeric assembly, is without precedent. The significance of our findings to the architecture of the holoenzyme and the clamp-assembly apparatus of all other organisms is discussed.
Figures



References
-
- Cantor C.R. and Schimmel,P.R. (1980) Biophysical Chemistry Part I: The Conformation of Biological Macromolecules. W.H.Freeman, New York, NY, p. 137.
-
- Dallmann H.G. and McHenry,C.S. (1995) DnaX complex of Escherichia coli DNA polymerase III holoenzyme. Physical characterization of the DnaX subunits and complexes. J. Biol. Chem., 270, 29563–29569. - PubMed
-
- Dallmann H.G., Thimmig,R.L. and McHenry,C.S. (1995) DnaX complex of Escherichia coli DNA polymerase III holoenzyme. Central role of τ in initiation complex assembly and in determining the functional asymmetry of holoenzyme. J. Biol. Chem., 270, 29555–29562. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

