Adult spinal cord stem cells generate neurons after transplantation in the adult dentate gyrus
- PMID: 11102479
- PMCID: PMC6773057
- DOI: 10.1523/JNEUROSCI.20-23-08727.2000
Adult spinal cord stem cells generate neurons after transplantation in the adult dentate gyrus
Abstract
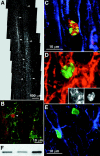
The adult rat spinal cord contains cells that can proliferate and differentiate into astrocytes and oligodendroglia in situ. Using clonal and subclonal analyses we demonstrate that, in contrast to progenitors isolated from the adult mouse spinal cord with a combination of growth factors, progenitors isolated from the adult rat spinal cord using basic fibroblast growth factor alone display stem cell properties as defined by their multipotentiality and self-renewal. Clonal cultures derived from single founder cells generate neurons, astrocytes, and oligodendrocytes, confirming the multipotent nature of the parent cell. Subcloning analysis showed that after serial passaging, recloning, and expansion, these cells retained multipotentiality, indicating that they are self-renewing. Transplantation of an in vitro-expanded clonal population of cells into the adult rat spinal cord resulted in their differentiation into glial cells only. However, after heterotopic transplantation into the hippocampus, transplanted cells that integrated in the granular cell layer differentiated into cells characteristic of this region, whereas engraftment into other hippocampal regions resulted in the differentiation of cells with astroglial and oligodendroglial phenotypes. The data indicate that clonally expanded, multipotent adult progenitor cells from a non-neurogenic region are not lineage-restricted to their developmental origin but can generate region-specific neurons in vivo when exposed to the appropriate environmental cues.
Figures






References
-
- Adrian EK, Jr, Walker BE. Incorporation of thymidine-3H by cells in normal and injured mouse spinal cord. J Neuropathol Exp Neurol. 1962;21:597–609. - PubMed
-
- Altman J, Bayer SA. The development of the rat spinal cord. Adv Anat Embryol Cell Biol. 1984;85:1–164. - PubMed
-
- Baimbridge KG, Miller JJ. Immunohistochemical localization of calcium-binding protein in the cerebellum, hippocampal formation and olfactory bulb of the rat. Brain Res. 1982;245:223–229. - PubMed
-
- Bayer SA, Yakel SW, Puri PS. Neurons in the rat dentate gyrus granular layer substantially increase during juvenile and adult life. Science. 1982;216:890–892. - PubMed
-
- Bhat RV, Axt KJ, Fosnaugh JS, Smith KJ, Johnson KA, Hill DE, Kinzler KW, Baraban JM. Expression of the APC tumor suppressor protein in oligodendroglia. Glia. 1996;17:169–174. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
