Adenosine kinase of Arabidopsis. Kinetic properties and gene expression
- PMID: 11115893
- PMCID: PMC59874
- DOI: 10.1104/pp.124.4.1775
Adenosine kinase of Arabidopsis. Kinetic properties and gene expression
Abstract
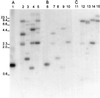
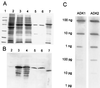
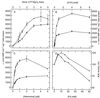
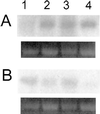
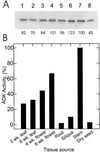
To assess the functional significance of adenosine salvage in plants, the cDNAs and genes encoding two isoforms of adenosine kinase (ADK) were isolated from Arabidopsis. The ADK1- and ADK2-coding sequences are very similar, sharing 92% and 89% amino acid and nucleotide identity, respectively. Each cDNA was overexpressed in Escherichia coli, and the catalytic activity of each isoform was determined. Both ADKs had similar catalytic properties with a K(m) and V(max)/K(m) for adenosine of 0.3 to 0.5 microM and 5.4 to 22 L min(-1) mg(-1) protein, respectively. The K(m) and V(max)/K(m) for the cytokinin riboside N(6)(isopentenyl) adenosine are 3 to 5 microM and 0.021 to 0.14 L min(-1) mg(-1) protein, respectively, suggesting that adenosine is the preferred substrate for both ADK isoforms. In Arabidopsis plants, both ADK genes are expressed constitutively, with the highest steady-state mRNA levels being found in stem and root. ADK1 transcript levels were generally higher than those of ADK2. ADK enzyme activity reflected relative ADK protein levels seen in immunoblots for leaves, flowers, and stems but only poorly so for roots, siliques, and dry seeds. The catalytic properties, tissue accumulation, and expression levels of these ADKs suggest that they play a key metabolic role in the salvage synthesis of adenylates and methyl recycling in Arabidopsis. They may also contribute to cytokinin interconversion.
Figures
References
-
- Åstot C, Dolczal K, Moritz T, Sandberg G. Precolumn derivatization and capillary liquid chromatography/frit-fast atom bombardment mass spectrometric analysis of cytokinins in Arabidopsis thaliana. J Mass Spectrom. 1998;33:892–902. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

