The spliceosome deposits multiple proteins 20-24 nucleotides upstream of mRNA exon-exon junctions
- PMID: 11118221
- PMCID: PMC305905
- DOI: 10.1093/emboj/19.24.6860
The spliceosome deposits multiple proteins 20-24 nucleotides upstream of mRNA exon-exon junctions
Abstract
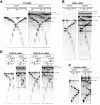
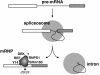
Eukaryotic mRNAs exist in vivo as ribonucleoprotein particles (mRNPs). The protein components of mRNPs have important functions in mRNA metabolism, including effects on subcellular localization, translational efficiency and mRNA half-life. There is accumulating evidence that pre-mRNA splicing can alter mRNP structure and thereby affect downstream mRNA metabolism. Here, we report that the spliceosome stably deposits several proteins on mRNAs, probably as a single complex of approximately 335 kDa. This complex protects 8 nucleotides of mRNA from complete RNase digestion at a conserved position 20-24 nucleotides upstream of exon-exon junctions. Splicing-dependent RNase protection of this region was observed in both HeLa cell nuclear extracts and Xenopus laevis oocyte nuclei. Immunoprecipitations revealed that five components of the complex are the splicing-associated factors SRm160, DEK and RNPS1, the mRNA-associated shuttling protein Y14 and the mRNA export factor REF. Possible functions for this complex in nucleocytoplasmic transport of spliced mRNA, as well as the nonsense-mediated mRNA decay pathway, are discussed.
Figures


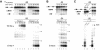

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

