Purification and kinetic analysis of recombinant CA XII, a membrane carbonic anhydrase overexpressed in certain cancers
- PMID: 11121027
- PMCID: PMC18897
- DOI: 10.1073/pnas.97.26.14212
Purification and kinetic analysis of recombinant CA XII, a membrane carbonic anhydrase overexpressed in certain cancers
Abstract
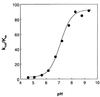
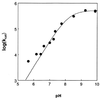
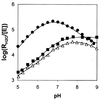
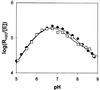
Carbonic anhydrase XII (CA XII) is a transmembrane glycoprotein with an active extracellular CA domain that is overexpressed on cell surfaces of certain cancers. Its expression has been linked to tumor invasiveness. To characterize its catalytic properties, we purified recombinant secretory forms of wild-type and mutant CA XIIs. The catalytic properties of these enzymes in the hydration of CO(2) were measured at steady state by stopped-flow spectrophotometry and at chemical equilibrium by the exchange of (18)O between CO(2) and water determined by mass spectrometry. The catalysis of CO(2) hydration by soluble CA XII has a maximal value of k(cat)/K(m) at 34 microM(-1) small middle dots(-1), which is similar to those of the membrane-associated CA IV and to soluble CA I. The pH profiles of this catalysis and the catalyzed hydrolysis of 4-nitrophenylacetate indicate that the pK(a) of the zinc-bound water in CA XII is 7.1. His64 in CA XII acts as a proton shuttle residue, as evidenced by the reduced rate constant for proton transfer in the mutants containing the replacements His64 --> Ala and His64 --> Arg, as well as by the selective inhibition of the proton transfer step by cupric ions in wild-type CA XII. The catalytic rate of CO(2) hydration by the soluble form of CA XII is identical with that of the membrane-bound enzyme. These observations suggest a role for CA XII in CO(2)/HCO(3)(-) homeostasis in cells in which it is normally expressed. They are also compatible with a role for CA XII in acidifying the microenvironment of cancer cells in which CA XII is overexpressed, providing a mechanism for CA XII to augment tumor invasiveness and suggesting CA XII as a potential target for chemotherapeutic agents.
Figures


Similar articles
-
The catalytic properties of murine carbonic anhydrase VII.Biochemistry. 1998 Jul 28;37(30):10837-45. doi: 10.1021/bi980046t. Biochemistry. 1998. PMID: 9692974
-
Glutamate and aspartate as proton shuttles in mutants of carbonic anhydrase.Biochemistry. 1997 Dec 16;36(50):15758-64. doi: 10.1021/bi972081q. Biochemistry. 1997. PMID: 9398305
-
Structure-based design of an intramolecular proton transfer site in murine carbonic anhydrase V.Biochemistry. 1996 Sep 10;35(36):11605-11. doi: 10.1021/bi9608018. Biochemistry. 1996. PMID: 8794740
-
The cellular physiology of carbonic anhydrases.JOP. 2001 Jul;2(4 Suppl):159-64. JOP. 2001. PMID: 11875253 Review.
-
Designing of novel carbonic anhydrase inhibitors and activators.Curr Med Chem Cardiovasc Hematol Agents. 2004 Jan;2(1):49-68. Curr Med Chem Cardiovasc Hematol Agents. 2004. PMID: 15328829 Review.
Cited by
-
Switching the Inhibitor-Enzyme Recognition Profile via Chimeric Carbonic Anhydrase XII.ChemistryOpen. 2021 May;10(5):567-580. doi: 10.1002/open.202100042. ChemistryOpen. 2021. PMID: 33945229 Free PMC article.
-
Expression of hypoxia-inducible carbonic anhydrases in brain tumors.Neuro Oncol. 2005 Oct;7(4):465-75. doi: 10.1215/S1152851705000025. Neuro Oncol. 2005. PMID: 16212811 Free PMC article.
-
Generation and characterization of the first inhibitory antibody targeting tumour-associated carbonic anhydrase XII.Cancer Immunol Immunother. 2011 May;60(5):649-58. doi: 10.1007/s00262-011-0980-z. Epub 2011 Feb 5. Cancer Immunol Immunother. 2011. PMID: 21298264 Free PMC article.
-
Glycosylation-independent targeting enhances enzyme delivery to lysosomes and decreases storage in mucopolysaccharidosis type VII mice.Proc Natl Acad Sci U S A. 2004 Mar 2;101(9):3083-8. doi: 10.1073/pnas.0308728100. Epub 2004 Feb 19. Proc Natl Acad Sci U S A. 2004. PMID: 14976248 Free PMC article.
-
Carbonic anhydrases in normal gastrointestinal tract and gastrointestinal tumours.World J Gastroenterol. 2005 Jan 14;11(2):155-63. doi: 10.3748/wjg.v11.i2.155. World J Gastroenterol. 2005. PMID: 15633208 Free PMC article. Review.
References
-
- Torczynski, R. M. & Bollon, A. P. (1996) U.S. Patent 5,589,579.
-
- Latif F, Tory K, Gnarra J, Yao M, Duh F M, Orcutt M L, Stackhouse T, Kuzmin I, Modi W, Geil L, et al. Science. 1993;260:1317–1320. - PubMed
-
- Chegwidden W R, Spencer I M. Inflammopharmacology. 1995;3:231–239.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

