Flightin is essential for thick filament assembly and sarcomere stability in Drosophila flight muscles
- PMID: 11134077
- PMCID: PMC2150682
- DOI: 10.1083/jcb.151.7.1483
Flightin is essential for thick filament assembly and sarcomere stability in Drosophila flight muscles
Abstract
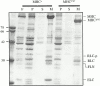
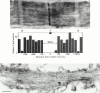
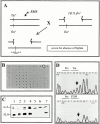
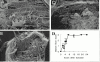
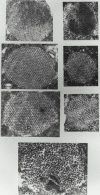
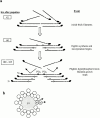
Flightin is a multiply phosphorylated, 20-kD myofibrillar protein found in Drosophila indirect flight muscles (IFM). Previous work suggests that flightin plays an essential, as yet undefined, role in normal sarcomere structure and contractile activity. Here we show that flightin is associated with thick filaments where it is likely to interact with the myosin rod. We have created a null mutation for flightin, fln(0), that results in loss of flight ability but has no effect on fecundity or viability. Electron microscopy comparing pupa and adult fln(0) IFM shows that sarcomeres, and thick and thin filaments in pupal IFM, are 25-30% longer than in wild type. fln(0) fibers are abnormally wavy, but sarcomere and myotendon structure in pupa are otherwise normal. Within the first 5 h of adult life and beginning of contractile activity, IFM fibers become disrupted as thick filaments and sarcomeres are variably shortened, and myofibrils are ruptured at the myotendon junction. Unusual empty pockets and granular material interrupt the filament lattice of adult fln(0) sarcomeres. Site-specific cleavage of myosin heavy chain occurs during this period. That myosin is cleaved in the absence of flightin is consistent with the immunolocalization of flightin on the thick filament and biochemical and genetic evidence suggesting it is associated with the myosin rod. Our results indicate that flightin is required for the establishment of normal thick filament length during late pupal development and thick filament stability in adult after initiation of contractile activity.
Figures






Similar articles
-
Flight muscle properties and aerodynamic performance of Drosophila expressing a flightin transgene.J Exp Biol. 2005 Feb;208(Pt 3):549-60. doi: 10.1242/jeb.01425. J Exp Biol. 2005. PMID: 15671343
-
COOH-terminal truncation of flightin decreases myofilament lattice organization, cross-bridge binding, and power output in Drosophila indirect flight muscle.Am J Physiol Cell Physiol. 2011 Aug;301(2):C383-91. doi: 10.1152/ajpcell.00016.2011. Epub 2011 May 18. Am J Physiol Cell Physiol. 2011. PMID: 21593450 Free PMC article.
-
Alterations in flightin phosphorylation in Drosophila flight muscles are associated with myofibrillar defects engendered by actin and myosin heavy-chain mutant alleles.Biochem Genet. 1994 Aug;32(7-8):301-14. doi: 10.1007/BF00555832. Biochem Genet. 1994. PMID: 7826316
-
Myosin: Formation and maintenance of thick filaments.Anim Sci J. 2019 Jul;90(7):801-807. doi: 10.1111/asj.13226. Epub 2019 May 27. Anim Sci J. 2019. PMID: 31134719 Free PMC article. Review.
-
Determining structure/function relationships for sarcomeric myosin heavy chain by genetic and transgenic manipulation of Drosophila.Microsc Res Tech. 2000 Sep 15;50(6):430-42. doi: 10.1002/1097-0029(20000915)50:6<430::AID-JEMT2>3.0.CO;2-E. Microsc Res Tech. 2000. PMID: 10998634 Review.
Cited by
-
An evolutionary analysis of orphan genes in Drosophila.Genome Res. 2003 Oct;13(10):2213-9. doi: 10.1101/gr.1311003. Genome Res. 2003. PMID: 14525923 Free PMC article.
-
Spalt mediates an evolutionarily conserved switch to fibrillar muscle fate in insects.Nature. 2011 Nov 16;479(7373):406-9. doi: 10.1038/nature10559. Nature. 2011. PMID: 22094701
-
Contiguity and Structural Impacts of a Non-Myosin Protein within the Thick Filament Myosin Layers.Biology (Basel). 2021 Jul 2;10(7):613. doi: 10.3390/biology10070613. Biology (Basel). 2021. PMID: 34356468 Free PMC article.
-
Molecular Characterization and Phylogenetic Analysis of Flightin Gene in Vespa basalis (Hymenoptera, Vespidae).Animals (Basel). 2024 Mar 21;14(6):978. doi: 10.3390/ani14060978. Animals (Basel). 2024. PMID: 38540074 Free PMC article.
-
An evolutionary analysis of flightin reveals a conserved motif unique and widespread in Pancrustacea.J Mol Evol. 2014 Jan;78(1):24-37. doi: 10.1007/s00239-013-9597-5. Epub 2013 Nov 23. J Mol Evol. 2014. PMID: 24271855
References
-
- Barral J.M., Epstein H.F. Protein machines and self assembly in muscle organization. BioEssays. 1999;21:813–823. - PubMed
-
- Beall C.J., Sepanski M.A., Fyrberg E.A. Genetic dissection of Drosophila myofibril formationeffects of actin and myosin heavy chain null alleles. Genes Dev. 1989;3:131–140. - PubMed
-
- Beinbrech G., Ashton F.T., Pepe F. The invertebrate myosin filamentsubfilament arrangement in the wall of tubular filaments of insect flight muscles. J. Mol. Biol. 1988;201:557–565. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

