Role of CD28/CD80-86 and CD40/CD154 costimulatory interactions in host defense to primary herpes simplex virus infection
- PMID: 11134274
- PMCID: PMC113957
- DOI: 10.1128/JVI.75.2.612-621.2001
Role of CD28/CD80-86 and CD40/CD154 costimulatory interactions in host defense to primary herpes simplex virus infection
Abstract
Dependence of the primary antiviral immune response on costimulatory interactions between CD28/CD80-86 and between CD40/CD154 (CD40 ligand) has been correlated with the extent of viral replication in two models of systemic infection, lymphocytic choriomeningitis virus and vesicular stomatitis virus. To determine the role of these costimulatory interactions in the context of an acute cytolytic, but locally replicating viral infection, herpes simplex virus (HSV) infection was assessed in mice that had the CD28/CD80-86 or CD40/CD154 interactions disrupted either genetically or with blocking reagents (CTLA4Ig and MR1, respectively). CTLA4Ig treatment greatly reduced paralysis-free survival during primary acute HSV infection. This reflected an almost total ablation of the anti-HSV CD4(+) and CD8(+) T-cell responses due to anergy and reduced cell numbers, respectively. Disruption of CD40/CD154 interactions impaired survival, but the effect was less severe than that observed in CTLA4Ig-treated mice, with reductions observed in the CD4(+) T-cell but not CD8(+) T-cell responses. These two costimulatory pathways functioned in part independently, since disruption of both further impaired survival. The dependence on these costimulatory interactions for the control of primary HSV infection may represent a more widespread paradigm for nonsystemic viruses, which have restricted sites of replication and which employ immunoevasive measures.
Figures


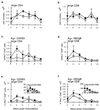
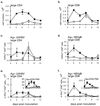

 ), CTLA4Ig-treated mice (■), and corresponding controls (
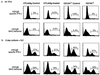
), CTLA4Ig-treated mice (■), and corresponding controls ( ) were analyzed directly ex vivo or after 3 days in culture with or without IL-2 (5 ng/ml) for HSV-specific IFN-γ production in response to UVHSV or HSVgB498–505. Plots represent fractions of IFN-γ-producing HSV-specific CD4+ or CD8+ cells in large CD4+ or CD8+ populations in CD154−/− (a) or CTLA4Ig-treated (b) mice.
) were analyzed directly ex vivo or after 3 days in culture with or without IL-2 (5 ng/ml) for HSV-specific IFN-γ production in response to UVHSV or HSVgB498–505. Plots represent fractions of IFN-γ-producing HSV-specific CD4+ or CD8+ cells in large CD4+ or CD8+ populations in CD154−/− (a) or CTLA4Ig-treated (b) mice.

Similar articles
-
Different costimulation signals used by CD4(+) and CD8(+) cells that independently initiate rejection of allogenic hepatocytes in mice.Hepatology. 2000 Nov;32(5):1018-28. doi: 10.1053/jhep.2000.19325. Hepatology. 2000. PMID: 11050052
-
[Influence of blocking B7/CD28 and CD40/CD154 co-stimulatory signals on immune function of sensitized mice].Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2014 Jun;22(3):801-6. doi: 10.7534/j.issn.1009-2137.2014.03.042. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2014. PMID: 24989298 Chinese.
-
Blocking CD40 - CD154 and CD80/CD86 - CD28 interactions during primary allogeneic stimulation results in T cell anergy and high IL-10 production.Eur J Immunol. 1999 Aug;29(8):2367-75. doi: 10.1002/(SICI)1521-4141(199908)29:08<2367::AID-IMMU2367>3.0.CO;2-3. Eur J Immunol. 1999. PMID: 10458748
-
The development of CD4+ T effector cells during the type 2 immune response.Immunol Res. 1999;20(1):55-65. doi: 10.1007/BF02786507. Immunol Res. 1999. PMID: 10467983 Review.
-
Targeting the CD40-CD154 Signaling Pathway for Treatment of Autoimmune Arthritis.Cells. 2019 Aug 18;8(8):927. doi: 10.3390/cells8080927. Cells. 2019. PMID: 31426619 Free PMC article. Review.
Cited by
-
Preferential use of B7.2 and not B7.1 in priming of vaccinia virus-specific CD8 T cells.J Immunol. 2009 Mar 1;182(5):2909-18. doi: 10.4049/jimmunol.0803545. J Immunol. 2009. PMID: 19234186 Free PMC article.
-
Differential requirements for CD80/86-CD28 costimulation in primary and memory CD4 T cell responses to vaccinia virus.Cell Immunol. 2011;266(2):130-4. doi: 10.1016/j.cellimm.2010.09.008. Epub 2010 Oct 30. Cell Immunol. 2011. PMID: 21040905 Free PMC article.
-
Multiple layers of CD80/86-dependent costimulatory activity regulate primary, memory, and secondary lymphocytic choriomeningitis virus-specific T cell immunity.J Virol. 2012 Feb;86(4):1955-70. doi: 10.1128/JVI.05949-11. Epub 2011 Dec 7. J Virol. 2012. PMID: 22156513 Free PMC article.
-
B7 costimulation molecules expressed from the herpes simplex virus 2 genome rescue immune induction in B7-deficient mice.J Virol. 2007 Nov;81(22):12200-9. doi: 10.1128/JVI.01224-07. Epub 2007 Sep 5. J Virol. 2007. PMID: 17804511 Free PMC article.
-
Control of memory CD8+ T cell differentiation by CD80/CD86-CD28 costimulation and restoration by IL-2 during the recall response.J Immunol. 2008 Jan 15;180(2):1148-57. doi: 10.4049/jimmunol.180.2.1148. J Immunol. 2008. PMID: 18178855 Free PMC article.
References
-
- Adler H, Beland J L, Del-Pan N C, Kobzik L, Sobel R A, Rimm I J. In the absence of T cells, natural killer cells protect from mortality due to HSV-1 encephalitis. J Neuroimmunol. 1999;93:208–213. - PubMed
-
- Andreasen S O, Christensen J E, Marker O, Thomsen A R. Role of CD40 ligand and CD28 in induction and maintenance of antiviral CD8+ effector T cell responses. J Immunol. 2000;164:3689–397. - PubMed
-
- Appleman L J, Berezovskaya A, Grass I, Boussiotis V A. CD28 costimulation mediates T cell expansion via IL-2-independent and IL-2-dependent regulation of cell cycle progression. J Immunol. 2000;164:144–151. - PubMed
-
- Bachmann M F, McKall-Faienza K, Schmits R, Bouchard D, Beach J, Speiser D E, Mak T W, Ohashi P S. Distinct roles for LFA-1 and CD28 during activation of naive T cells: adhesion versus costimulation. Immunity. 1997;7:549–557. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

