Mice devoid of fer protein-tyrosine kinase activity are viable and fertile but display reduced cortactin phosphorylation
- PMID: 11134346
- PMCID: PMC86629
- DOI: 10.1128/MCB.21.2.603-613.2001
Mice devoid of fer protein-tyrosine kinase activity are viable and fertile but display reduced cortactin phosphorylation
Abstract
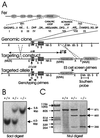
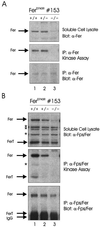
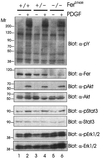
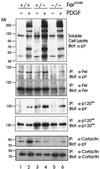
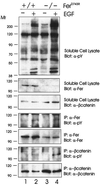
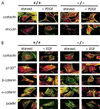
The ubiquitous Fer protein-tyrosine kinase has been proposed to regulate diverse processes such as cell growth, cell adhesion, and neurite outgrowth. To gain insight into the biological function of Fer, we have targeted the fer locus with a kinase-inactivating missense mutation (fer(D743R)). Mice homozygous for this mutation develop normally, have no overt phenotypic differences from wild-type mice, and are fertile. Since these mice lack both Fer and the testis-specific FerT kinase activities, these proteins are clearly not essential for development and survival. No differences were observed in overall cellularity of bone marrow, spleen, or thymus in the absence of Fer activity. While most platelet-derived growth factor (PDGF)-induced tyrosine phosphorylation was unchanged in fer(D743R) homozygous embryonic fibroblasts, cortactin phosphorylation was reduced. However, Fer kinase activity was not required for PDGF-induced Stat3, p120(ctn), or epidermal growth factor (EGF)-induced beta-catenin phosphorylation. Also, no defects were observed in changes to the actin cytoskeleton, adherens junctions, or focal adhesions in PDGF- or EGF-stimulated fer(D743R) homozygous embryonic fibroblasts. Therefore, Fer likely serves a redundant role in regulating cell growth, cell adhesion, retinal development, and spermatogenesis but is required for efficient phosphorylation of cortactin.
Figures
Similar articles
-
Cell volume-dependent phosphorylation of proteins of the cortical cytoskeleton and cell-cell contact sites. The role of Fyn and FER kinases.J Biol Chem. 2000 Oct 13;275(41):32289-98. doi: 10.1074/jbc.M003172200. J Biol Chem. 2000. PMID: 10921917
-
Continuous association of cadherin with beta-catenin requires the non-receptor tyrosine-kinase Fer.J Cell Sci. 2004 Jul 1;117(Pt 15):3207-19. doi: 10.1242/jcs.01174. J Cell Sci. 2004. PMID: 15226396
-
p120 Catenin-associated Fer and Fyn tyrosine kinases regulate beta-catenin Tyr-142 phosphorylation and beta-catenin-alpha-catenin Interaction.Mol Cell Biol. 2003 Apr;23(7):2287-97. doi: 10.1128/MCB.23.7.2287-2297.2003. Mol Cell Biol. 2003. PMID: 12640114 Free PMC article.
-
Role of c-Src tyrosine kinase in EGF-induced mitogenesis.Front Biosci. 1997 Oct 15;2:d501-18. doi: 10.2741/a208. Front Biosci. 1997. PMID: 9331427 Review.
-
Tyrosine protein kinases and spermatogenesis: truncation matters.Mol Reprod Dev. 2006 Apr;73(4):399-403. doi: 10.1002/mrd.20456. Mol Reprod Dev. 2006. PMID: 16432822 Review.
Cited by
-
Transient Sperm Starvation Improves the Outcome of Assisted Reproductive Technologies.Front Cell Dev Biol. 2019 Nov 5;7:262. doi: 10.3389/fcell.2019.00262. eCollection 2019. Front Cell Dev Biol. 2019. PMID: 31750304 Free PMC article.
-
Absence of Fer protein tyrosine kinase exacerbates endotoxin induced intestinal epithelial barrier dysfunction in vivo.Gut. 2005 Aug;54(8):1091-7. doi: 10.1136/gut.2004.061887. Gut. 2005. PMID: 16009680 Free PMC article.
-
Identification of Fer tyrosine kinase localized on microtubules as a platelet endothelial cell adhesion molecule-1 phosphorylating kinase in vascular endothelial cells.Mol Biol Cell. 2003 Sep;14(9):3553-64. doi: 10.1091/mbc.e03-02-0080. Epub 2003 Jun 13. Mol Biol Cell. 2003. PMID: 12972546 Free PMC article.
-
Feline sarcoma-related protein expression correlates with malignant aggressiveness and poor prognosis in renal cell carcinoma.Cancer Sci. 2013 Jun;104(6):681-6. doi: 10.1111/cas.12140. Epub 2013 Apr 18. Cancer Sci. 2013. PMID: 23445469 Free PMC article.
-
The Fer tyrosine kinase regulates an axon retraction response to Semaphorin 3A in dorsal root ganglion neurons.BMC Dev Biol. 2007 Nov 30;7:133. doi: 10.1186/1471-213X-7-133. BMC Dev Biol. 2007. PMID: 18053124 Free PMC article.
References
-
- Alcalay M, Antolini F, Van de Ven W J, Lanfrancone L, Grignani F, Pelicci P G. Characterization of human and mouse c-fes cDNA clones and identification of the 5′ end of the gene. Oncogene. 1990;5:267–275. - PubMed
-
- Calautti E, Missero C, Stein P L, Ezzell R M, Dotto G P. Fyn tyrosine kinase is involved in keratinocyte differentiation control. Genes Dev. 1995;9:2279–2291. - PubMed
-
- Care A, Mattia G, Montesoro E, Parolini I, Russo G, Colombo M P, Peschle C. c-fes expression in ontogenetic development and hematopoietic differentiation. Oncogene. 1994;9:739–747. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
