Prophylactic treatment of migraine with angiotensin converting enzyme inhibitor (lisinopril): randomised, placebo controlled, crossover study
- PMID: 11141144
- PMCID: PMC26600
- DOI: 10.1136/bmj.322.7277.19
Prophylactic treatment of migraine with angiotensin converting enzyme inhibitor (lisinopril): randomised, placebo controlled, crossover study
Abstract
Objective: To determine the efficacy of an angiotensin converting enzyme inhibitor in the prophylaxis of migraine.
Design: Double blind, placebo controlled, crossover study.
Setting: Neurological outpatient clinic.
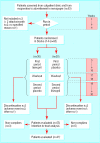
Participants: Sixty patients aged 19-59 years with migraine with two to six episodes a month.
Interventions: Treatment period of 12 weeks with one 10 mg lisinopril tablet once daily for one week then two 10 mg lisinopril tablets once daily for 11 weeks, followed by a two week wash out period. Second treatment period of one placebo tablet once daily for one week and then two placebo tablets for 11 weeks. Thirty participants followed this schedule, and 30 received placebo followed by lisinopril.
Main outcome measures: Primary end points: number of hours with headache, number of days with headache, number of days with migraine. Secondary end points: headache severity index, use of drugs for symptomatic relief, quality of life and number of days taken as sick leave, acceptability of treatment.
Results: In the 47 participants with complete data, hours with headache, days with headache, days with migraine, and headache severity index were significantly reduced by 20% (95% confidence interval 5% to 36%), 17% (5% to 30%), 21% (9% to 34%), and 20% (3% to 37%), respectively, with lisinopril compared with placebo. Days with migraine were reduced by at least 50% in 14 participants for active treatment versus placebo and 17 patients for active treatment versus run-in period. Days with migraine were fewer by at least 50% in 14 participants for active treatment versus placebo. Intention to treat analysis of data from 55 patients supported the differences in favour of lisinopril for the primary end points.
Conclusion: The angiotensin converting enzyme inhibitor, lisinopril, has a clinically important prophylactic effect in migraine.
Figures
References
-
- Bender WI. ACE inhibitors for prophylaxis of migraine headaches. Headache. 1995;35:470–471. - PubMed
-
- International Headache Society Committee on Clinical Trials in Migraine. Guidelines for controlled trials of drugs in migraine. Cephalalgia. 1991;11/1:1–12. - PubMed
-
- Headache Classification Committee of the International Headache Society. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia. 1988;8(suppl 7):1–96. - PubMed
-
- Ware JE, Gandek B IQOLA project group. The SF-36 health survey: development and use in mental health research and the IQOLA project. Int J Mental Health. 1994;23:49–73.
-
- Lachin JM. Introduction to sample size determination and power analysis in clinical trials. Control Clin Trials. 1981;2:93–113. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical