Cerebral malaria in mice: interleukin-2 treatment induces accumulation of gammadelta T cells in the brain and alters resistant mice to susceptible-like phenotype
- PMID: 11141489
- PMCID: PMC1850276
- DOI: 10.1016/s0002-9440(10)63954-5
Cerebral malaria in mice: interleukin-2 treatment induces accumulation of gammadelta T cells in the brain and alters resistant mice to susceptible-like phenotype
Abstract
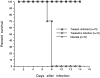
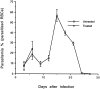
In this study, we report that infection with Plasmodium yoelii 17XL, a lethal strain of rodent malaria, does not result in death in the DBA/2 strain of mice. In contrast to BALB/c mice, DBA/2 mice developed significantly less parasitemia and never manifested symptoms of cerebral malaria (CM) on infection with this parasite. Moreover, the histological changes evident in the brain of susceptible BALB/c were absent in DBA/2 mice. Interestingly, the resistant DBA/2 mice when treated with recombinant interleukin (IL)-2, were found to develop CM symptoms and the infection became fatal by 6 to 8 days after infection. This condition was associated with an augmented interferon-gamma and nitric oxide production. Unexpectedly, IL-10 levels were also elevated in IL-2-treated DBA/2 mice during late stage of infection (at day 6 of infection) whereas the inverse relationship between IL-10 and interferon-gamma or nitric oxide was maintained in the early stage of infection (at day 3 after infection). The level of tumor necrosis factor-alpha production was moderately increased in the late phase of infection in these mice. Histology of brain from IL-2-treated mice demonstrated the presence of parasitized erythrocytes and infiltration of lymphocytes in cerebral vessels, and also displayed some signs of endothelial degeneration. Confocal microscopical studies demonstrated preferential accumulation of gammadelta T cells in the cerebral vessels of IL-2-treated and -infected mice but not in mice treated with IL-2 alone. The cells recruited in the brain were activated because they demonstrated expression of CD25 (IL-2R) and CD54 (intercellular adhesion molecule 1) molecules. Administration of anti-gammadelta mAb prevented development of CM in IL-2-treated mice until day 18 after infection whereas mice treated with control antibody showed CM symptoms by day 6 after infection. The information concerning creating pathological sequelae and death in an otherwise resistant mouse strain provides an interesting focus for the burden of pathological attributes on death in an infectious disease.
Figures
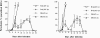



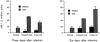

Similar articles
-
Reasons why DBA/2 mice are resistant to malarial infection: expansion of CD3int B220+ gammadelta T cells with double-negative CD4- CD8- phenotype in the liver.Immunology. 2006 Jan;117(1):127-35. doi: 10.1111/j.1365-2567.2005.02273.x. Immunology. 2006. PMID: 16423048 Free PMC article.
-
Correlation of increased expression of intercellular adhesion molecule-1, but not high levels of tumor necrosis factor-alpha, with lethality of Plasmodium yoelii 17XL, a rodent model of cerebral malaria.Am J Trop Med Hyg. 1998 Dec;59(6):852-8. doi: 10.4269/ajtmh.1998.59.852. Am J Trop Med Hyg. 1998. PMID: 9886187
-
Effects of in vivo administration of anti-IL-10 or anti-IFN-gamma monoclonal antibody on the host defense mechanism against Plasmodium yoelii yoelii infection.J Vet Med Sci. 2000 Jun;62(6):583-7. doi: 10.1292/jvms.62.583. J Vet Med Sci. 2000. PMID: 10907683
-
Tumor necrosis factor alpha in the pathogenesis of cerebral malaria.Cell Mol Life Sci. 2003 Aug;60(8):1623-35. doi: 10.1007/s00018-003-2347-x. Cell Mol Life Sci. 2003. PMID: 14504653 Free PMC article. Review.
-
Cerebral malaria -- a neurovascular pathology with many riddles still to be solved.Curr Neurovasc Res. 2004 Apr;1(2):91-110. doi: 10.2174/1567202043480116. Curr Neurovasc Res. 2004. PMID: 16185187 Review.
Cited by
-
Plasmodium berghei ANKA infection increases Foxp3, IL-10 and IL-2 in CXCL-10 deficient C57BL/6 mice.Malar J. 2011 Mar 28;10:69. doi: 10.1186/1475-2875-10-69. Malar J. 2011. PMID: 21439091 Free PMC article.
-
Experimental cerebral malaria pathogenesis--hemodynamics at the blood brain barrier.PLoS Pathog. 2014 Dec 4;10(12):e1004528. doi: 10.1371/journal.ppat.1004528. eCollection 2014 Dec. PLoS Pathog. 2014. PMID: 25474413 Free PMC article.
-
Reasons why DBA/2 mice are resistant to malarial infection: expansion of CD3int B220+ gammadelta T cells with double-negative CD4- CD8- phenotype in the liver.Immunology. 2006 Jan;117(1):127-35. doi: 10.1111/j.1365-2567.2005.02273.x. Immunology. 2006. PMID: 16423048 Free PMC article.
-
Scanning electron microscopy of the neuropathology of murine cerebral malaria.Malar J. 2006 Nov 24;5:116. doi: 10.1186/1475-2875-5-116. Malar J. 2006. PMID: 17125519 Free PMC article.
-
γδ T Cells in Brain Homeostasis and Diseases.Front Immunol. 2022 May 26;13:886397. doi: 10.3389/fimmu.2022.886397. eCollection 2022. Front Immunol. 2022. PMID: 35693762 Free PMC article. Review.
References
-
- Oaks SC, Mitchell VS, Pearson GW, Carpenter CJ (Eds): Malaria: Obstacles and Opportunities. Washington, DC, National Academy Press, 1991, pp 56–65 - PubMed
-
- Taylor-Robinson AW, Smith EC: A role for cytokines in potentiation of malaria vaccines through immunological modulation of blood stage infection. Immunol Rev 1999, 171:105-124 - PubMed
-
- Grau GE, Piguet PF, Engers HD, Louis JA, Vassalli P, Lambert PH: L3T4+ T lymphocytes play a major role in the pathogenesis of murine cerebral malaria. J Immunol 1986, 137:2348-2354 - PubMed
-
- Yanez DM, Manning DD, Cooley AJ, Weidanz WP, van der Heyde HC: Participation of lymphocyte subpopulations in the pathogenesis of experimental murine cerebral malaria. J Immunol 1996, 157:1620-1624 - PubMed
-
- Hermsen C, van de Wiel T, Mommers E, Sauerwein R, Eling W: Depletion of CD4+ or CD8+ T-cells prevents Plasmodium berghei induced cerebral malaria in end-stage disease. Parasitology 1997, 114:7-12 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

