Induction of heat shock protein 70 by herbimycin A and cyclopentenone prostaglandins in smooth muscle cells
- PMID: 11147963
- PMCID: PMC312898
- DOI: 10.1379/1466-1268(2000)005<0121:iohspb>2.0.co;2
Induction of heat shock protein 70 by herbimycin A and cyclopentenone prostaglandins in smooth muscle cells
Abstract
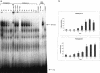
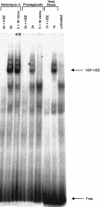
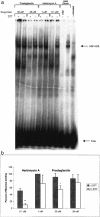
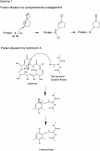
This study characterizes Hsp70 induction in human smooth muscle cells (SMC) by herbimycin A and cyclopentenone prostaglandins. The magnitude of Hsp70 induction by cyclopentenone prostaglandins was 8- to 10-fold higher than induction by herbimycin A. Hsp70 induction by delta12PGJ2 was first observed at 10 microM, rose to 4000-5000 ng/mL within one log unit and a maximum response was not observed; concentrations of delta12PGJ2 higher than 30 microM were toxic to the cells. A maximum response with herbimycin A (500 ng/mL) was reached at 0.05 microM and maintained to 1 microM without toxicity. Both, delta12PGJ2 and herbimycin A, were inhibited by dithiothreitol (DTT, 100 microM) at lower concentrations and became less sensitive to inhibition at higher concentrations. Hsp70 induction after incubation of SMC with delta12PGJ2 followed by addition of herbimycin A was significantly higher than Hsp70 induction after incubation with herbimycin A followed by addition of delta12PGJ2. When cells were incubated with [3H]-PGJ2, followed by protein denaturation, substantial radioactivity remained protein-bound suggesting that the prostaglandin must be covalently bound. Covalent binding was largely insensitive to DTT. Maximal Hsp70 induction was observed after 5 minutes of exposure of the cells to herbimycin A followed by a 20 hour recovery period in agent-free medium. Cells required 3-4 hours of exposure to delta12PGJ2 followed by a 20 hour recovery period in order to see high Hsp70 induction. Binding of the heat shock factor (HSF) to the heat shock element (HSE) in the presence of herbimycin A or delta12PGJ2, and the effects of DTT, mirrored the results of Hsp70 induction. The results suggest that probable differences between the 2 agents are at the level of the signal transduction prior to HSF activation.
Figures





References
-
- Benjamin IJ, McMillan DR. Stress (heat shock) proteins: molecular chaperones in cardiovascular biology and disease. Circ Res. 1998;83:117–132. - PubMed
-
- Conde AG, Lau SS, Dillmann WH, Mestril R. Induction of heat shock proteins by tyrosine kinase inhibitors in rat cardiomyocytes and myogenic cells confers protection against simulated ischemia. J Mol Cell Cardiol. 1997;26:1927–1938. - PubMed
-
- Feige U, Polla BS. Hsp70—a multi-gene, multi-structure, muti-function family with potential clinical applications. Experientia. 1994;50:979–986. - PubMed
-
- Fernandes M, O'Brien T, and Lis JT 1994 Structure and Regulation of Heat Shock Gene Promoters. In: The Biology of Heat Shock Proteins and Molecular Chaperones, eds Morimoto RI, Tissieres A, Georgopoulos C. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY , 375–393.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
