Wasp, the Drosophila Wiskott-Aldrich syndrome gene homologue, is required for cell fate decisions mediated by Notch signaling
- PMID: 11149916
- PMCID: PMC2193661
- DOI: 10.1083/jcb.152.1.1-b
Wasp, the Drosophila Wiskott-Aldrich syndrome gene homologue, is required for cell fate decisions mediated by Notch signaling
Abstract
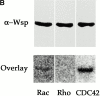
Wiskott-Aldrich syndrome proteins, encoded by the Wiskott-Aldrich syndrome gene family, bridge signal transduction pathways and the microfilament-based cytoskeleton. Mutations in the Drosophila homologue, Wasp (Wsp), reveal an essential requirement for this gene in implementation of cell fate decisions during adult and embryonic sensory organ development. Phenotypic analysis of Wsp mutant animals demonstrates a bias towards neuronal differentiation, at the expense of other cell types, resulting from improper execution of the program of asymmetric cell divisions which underlie sensory organ development. Generation of two similar daughter cells after division of the sensory organ precursor cell constitutes a prominent defect in the Wsp sensory organ lineage. The asymmetric segregation of key elements such as Numb is unaffected during this division, despite the misassignment of cell fates. The requirement for Wsp extends to additional cell fate decisions in lineages of the embryonic central nervous system and mesoderm. The nature of the Wsp mutant phenotypes, coupled with genetic interaction studies, identifies an essential role for Wsp in lineage decisions mediated by the Notch signaling pathway.
Figures









References
-
- Adams M.D., Celniker S.E., Holt R.A., Evans C.A., Gocayne J.D., Amanatides P.G., Scherer S.E., Li P.W., Hoskins R.A., Galle R.F. The genome sequence of Drosophila melanogaster . Science. 2000;287:2185–2195. - PubMed
-
- Ahmed Y., Hayashi S., Levine A., Wieschaus E. Regulation of armadillo by a Drosophila APC inhibits neuronal apoptosis during retinal development. Cell. 1998;93:1171–1182. - PubMed
-
- Artavanis-Tsakonas S., Rand M.D., Lake R.J. Notch signalingcell fate control and signal integration in development. Science. 1999;284:770–776. - PubMed
-
- Aspenstrom P., Lindberg U., Hall A. Two GTPases, Cdc42 and Rac, bind directly to a protein implicated in the immunodeficiency disorder Wiskott-Aldrich syndrome. Curr. Biol. 1996;6:70–75. - PubMed
-
- Bang A.G., Posakony J.W. The Drosophila gene Hairless encodes a novel basic protein that controls alternative cell fates in adult sensory organ development. Genes Dev. 1992;6:1752–1769. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

