Nerve injury induces a rapid efflux of nitric oxide (NO) detected with a novel NO microsensor
- PMID: 11150338
- PMCID: PMC6762443
- DOI: 10.1523/JNEUROSCI.21-01-00215.2001
Nerve injury induces a rapid efflux of nitric oxide (NO) detected with a novel NO microsensor
Abstract
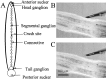
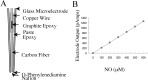
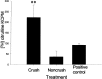
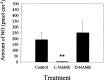
An early step in repair of the leech CNS is the appearance of endothelial nitric oxide synthase (eNOS) immunoreactivity and NOS activity, but coincident generation of NO at the lesion after injury has not been shown. This is important because NO can regulate microglial cell motility and axon growth. Indirect measurement of NO with the standard citrulline assay demonstrated that NO was generated within 30 min after nerve cord injury. A polarographic NO-selective self-referencing microelectrode that measures NO flux noninvasively was developed to obtain higher spatial and temporal resolution. With this probe, it was possible to demonstrate that immediately after the leech CNS was injured, NO left the lesion with a mean peak efflux of 803 +/- 99 fmol NO cm(-2) sec(-1). NO efflux exponentially declined to a constant value, as described through the equation f(t) = y(o) + ae(-t/tau), with tau = 117 +/- 30 sec. The constant y(o) = 15.8 +/- 4.5 fmol cm(-2) represents a sustained efflux of NO. Approximately 200 pmol NO cm(-2) is produced at the lesion (n = 8). Thus, injury activates eNOS already present in the CNS and precedes the accumulation of microglia at the lesion, consistent with the hypothesis that NO acts to stop the migrating microglia at the lesion site.
Figures

Similar articles
-
Nitric oxide influences injury-induced microglial migration and accumulation in the leech CNS.J Neurosci. 2000 Feb 1;20(3):1036-43. doi: 10.1523/JNEUROSCI.20-03-01036.2000. J Neurosci. 2000. PMID: 10648709 Free PMC article.
-
Injury-induced expression of endothelial nitric oxide synthase by glial and microglial cells in the leech central nervous system within minutes after injury.Proc Biol Sci. 1998 Nov 22;265(1411):2171-5. doi: 10.1098/rspb.1998.0555. Proc Biol Sci. 1998. PMID: 9872006 Free PMC article.
-
Methylene blue blocks cGMP production and disrupts directed migration of microglia to nerve lesions in the leech CNS.J Neurobiol. 2003 Nov;57(2):183-92. doi: 10.1002/neu.10262. J Neurobiol. 2003. PMID: 14556284
-
Repair of the central nervous system: lessons from lesions in leeches.J Neurobiol. 1995 Jul;27(3):353-66. doi: 10.1002/neu.480270308. J Neurobiol. 1995. PMID: 7673894 Review.
-
Modulation of acetylcholine release by nitric oxide.Prog Brain Res. 1996;109:219-24. doi: 10.1016/s0079-6123(08)62105-6. Prog Brain Res. 1996. PMID: 9009710 Review. No abstract available.
Cited by
-
Interaction of HmC1q with leech microglial cells: involvement of C1qBP-related molecule in the induction of cell chemotaxis.J Neuroinflammation. 2012 Feb 22;9:37. doi: 10.1186/1742-2094-9-37. J Neuroinflammation. 2012. PMID: 22356764 Free PMC article.
-
Arachidonic acid closes innexin/pannexin channels and thereby inhibits microglia cell movement to a nerve injury.Dev Neurobiol. 2013 Aug;73(8):621-31. doi: 10.1002/dneu.22088. Epub 2013 Jun 18. Dev Neurobiol. 2013. PMID: 23650255 Free PMC article.
-
Manganese Oxide Nanozymes Ameliorate Mechanical Allodynia in a Rat Model of Partial Sciatic Nerve-Transection Induced Neuropathic Pain.Int J Nanomedicine. 2019 Dec 27;14:10105-10117. doi: 10.2147/IJN.S225594. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31920306 Free PMC article.
-
Metabolic oxygen consumption measurement with a single-cell biosensor after particle microbeam irradiation.Radiat Environ Biophys. 2015 Mar;54(1):137-144. doi: 10.1007/s00411-014-0574-1. Epub 2014 Oct 22. Radiat Environ Biophys. 2015. PMID: 25335641 Free PMC article.
-
Expanding the question-answering potential of single-cell microbeams at RARAF, USA.J Radiat Res. 2009 Mar;50 Suppl A(Suppl A):A21-8. doi: 10.1269/jrr.08134s. J Radiat Res. 2009. PMID: 19346682 Free PMC article. Review.
References
-
- Balligand JL, Ungureanu-Longrois D, Simmons WW, Pimental D, Malinski TA, Kapturczak M, Taha Z, Lowenstein CJ, Davidoff AJ, Kelly RA, Smith TW, Michel T. Cytokine-inducible nitric oxide synthase (iNOS) expression in cardiac myocytes. J Biol Chem. 1994;269:27580–27588. - PubMed
-
- Banati RB, Gehrmann J, Schubert P, Kreutzberg GW. Cytotoxicity of microglia. Glia. 1993;7:111–118. - PubMed
-
- Brecknell JE, Fawcett JW. Axonal regeneration. Biol Rev Camb Philos Soc. 1996;71:227–255. - PubMed
-
- Bredt DS, Schmidt HH. The citrulline assay. In: Feelisch M, Stamler JS, editors. Methods in nitric oxide research. Wiley; New York: 1996. pp. 249–270.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
