Detection of diverse hepatitis C virus (HCV)-specific cytotoxic T lymphocytes in peripheral blood of infected persons by screening for responses to all translated proteins of HCV
- PMID: 11152496
- PMCID: PMC114029
- DOI: 10.1128/JVI.75.3.1229-1235.2001
Detection of diverse hepatitis C virus (HCV)-specific cytotoxic T lymphocytes in peripheral blood of infected persons by screening for responses to all translated proteins of HCV
Abstract
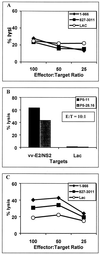
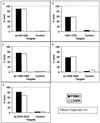
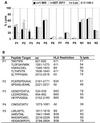
Broadly directed hepatitis C virus (HCV)-specific cytotoxic T lymphocytes (CTL) have been identified from liver-infiltrating lymphocytes but have been more difficult to assess in peripheral blood of infected persons. To enhance the detection of CTL from peripheral blood mononuclear cells (PBMC), we cocultured PBMC with autologous Epstein-Barr virus-transformed B-lymphoblastoid cell lines that had been infected with recombinant vaccinia virus constructs so that they expressed the entire translated polyprotein of HCV-H, a type 1a strain. These stimulated cells from HCV-infected as well as exposed seronegative persons were then cloned at limiting dilution and tested for HCV-specific CTL activity using a standard (51)Cr release assay. HCV-specific CTL were detected in PBMC from seven of nine persons with chronic hepatitis, including five of seven in whom CTL had previously been detected from liver biopsy specimens but not PBMC. In a single person with chronic HCV infection, CTL directed against as many as five different epitopes were detected in peripheral blood and were similar in specificity to those detected in liver tissue. This technique was used to evaluate eight subjects identified to be at high risk for HCV exposure due to continued injection drug abuse; no evidence of CTL in PBMC was found. We conclude that CTL can be detected in PBMC from the majority of persons with chronic HCV infection but are present at lower levels or absent in exposed but persistently seronegative persons. The high degree of concordance of HCV epitopes identified from liver and PBMC suggests that this strategy is a reasonable alternative to liver biopsy for characterizing the CTL response to HCV in chronically infected persons.
Figures
Similar articles
-
Characteristics of the intrahepatic cytotoxic T lymphocyte response in chronic hepatitis C virus infection.Springer Semin Immunopathol. 1997;19(1):69-83. doi: 10.1007/BF00945026. Springer Semin Immunopathol. 1997. PMID: 9266632 Review.
-
Liver-derived CTL in hepatitis C virus infection: breadth and specificity of responses in a cohort of persons with chronic infection.J Immunol. 1998 Feb 1;160(3):1479-88. J Immunol. 1998. PMID: 9570570
-
The role of hepatitis C virus-specific cytotoxic T lymphocytes in chronic hepatitis C.J Immunol. 1997 Feb 1;158(3):1473-81. J Immunol. 1997. PMID: 9013994
-
Effect of interferon-alpha therapy on epitope-specific cytotoxic T lymphocyte responses in hepatitis C virus-infected individuals.Eur J Immunol. 2002 Jan;32(1):144-54. doi: 10.1002/1521-4141(200201)32:1<144::AID-IMMU144>3.0.CO;2-X. Eur J Immunol. 2002. PMID: 11754355
-
Immunopathology of hepatitis C.Springer Semin Immunopathol. 1997;19(1):57-68. doi: 10.1007/BF00945025. Springer Semin Immunopathol. 1997. PMID: 9266631 Review.
Cited by
-
Virus-induced hepatocellular carcinomas cause antigen-specific local tolerance.J Clin Invest. 2013 Mar;123(3):1032-43. doi: 10.1172/JCI64742. Epub 2013 Feb 1. J Clin Invest. 2013. PMID: 23454765 Free PMC article.
-
Theileria annulata-transformed cell lines are efficient antigen-presenting cells for in vitro analysis of CD8 T cell responses to bovine herpesvirus-1.Vet Res. 2011 Dec 19;42(1):119. doi: 10.1186/1297-9716-42-119. Vet Res. 2011. PMID: 22182243 Free PMC article.
-
Simultaneous assessment of cytotoxic T lymphocyte responses against multiple viral infections by combined usage of optimal epitope matrices, anti- CD3 mAb T-cell expansion and "RecycleSpot".J Transl Med. 2005 May 11;3(1):20. doi: 10.1186/1479-5876-3-20. J Transl Med. 2005. PMID: 15888204 Free PMC article.
-
Dissection of two drug-targeted regions of Hepatitis C virus subtype 4a infecting Egyptian patients.Virus Genes. 2020 Oct;56(5):564-581. doi: 10.1007/s11262-020-01776-y. Epub 2020 Jun 22. Virus Genes. 2020. PMID: 32572756 Free PMC article.
-
Influence of host resistance on viral adaptation: hepatitis C virus as a case study.Infect Drug Resist. 2015 Apr 7;8:63-74. doi: 10.2147/IDR.S49891. eCollection 2015. Infect Drug Resist. 2015. PMID: 25897250 Free PMC article. Review.
References
-
- Alter M J, Kruszon-Moran D, Nainan O V, McQuillan G M, Gao F, Moyer L A, Kaslow R A, Margolis H S. The prevalence of hepatitis C virus infection in the United States, 1988 through 1994. N Engl J Med. 1999;341:556–562. - PubMed
-
- Battegay M, Fikes J, Di Bisceglie A M, Wentworth P A, Sette A, Celis E, Ching W M, Grakoui A, Rice C M, Kurokohchi K, et al. Patients with chronic hepatitis C have circulating cytotoxic T cells which recognize hepatitis C virus-encoded peptides binding to HLA-A2.1 molecules. J Virol. 1995;69:2462–2470. - PMC - PubMed
-
- Cerny A, Fowler P, Brothers M A, Houghton M, Schlicht H J, Chisari F V. Induction in vitro of a primary human antiviral cytotoxic T cell response. Eur J Immunol. 1995;25:627–630. - PubMed
-
- Choo Q L, Kuo G, Weiner A J, Overby L R, Bradley D W, Houghton M. Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome. Science. 1989;244:362–364. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

