Pre- and postjunctional protective effect of neocuproine on the nitrergic neurotransmitter in the mouse gastric fundus
- PMID: 11156587
- PMCID: PMC1572530
- DOI: 10.1038/sj.bjp.0703772
Pre- and postjunctional protective effect of neocuproine on the nitrergic neurotransmitter in the mouse gastric fundus
Abstract
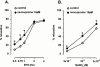
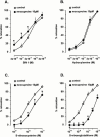
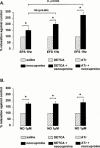
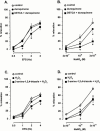
1. Electrical field stimulation (EFS) of non-adrenergic non-cholinergic nerves of the mouse gastric fundus induced frequency-dependent transient relaxations which were mimicked by nitric oxide (NO), added as acidified NaNO(2). The NO donors S-nitrosocysteine, S-nitrosoglutathione, SIN-1 and hydroxylamine induced sustained concentration-dependent relaxations. The NO synthase blocker L-nitro arginine (L-NOARG; 300 microM) abolished the relaxations to EFS without affecting the relaxations to NO. 2. The copper(I) chelator neocuproine (10 microM) enhanced the relaxations to EFS and NO but inhibited those to S-nitrosocysteine and S-nitrosoglutathione. Neocuproine potentiated the relaxations to SIN-1, which releases NO extracellularly, without affecting the relaxations to hydroxylamine, which releases NO intracellularly. 3. The potentiating effect of neocuproine on the relaxations to EFS was more pronounced after inhibition of catalase with 3-amino-1,2,4-triazole (1 mM) but not after inhibition of Cu/Zn superoxide dismutase (SOD) with diethyl dithiocarbamic acid (DETCA, 1 mM). The potentiating effect of neocuproine on relaxations to NO was not altered by 3-amino-1,2,4-triazole or DETCA treatment. 4. The relaxations to EFS were significantly inhibited by the oxidants hydrogen peroxide (70 microM) and duroquinone (10 microM) but only after inhibition of catalase with 3-amino-1,2,4-triazole or after inhibition of Cu/ZnSOD with DETCA respectively. 5. Our results suggest that neocuproine can act as an antioxidant in the mouse gastric fundus and that both catalase and Cu/ZnSOD protect the nitrergic neurotransmitter from oxidative breakdown. Since inhibition of catalase but not inhibition of Cu/ZnSOD potentiated the effect of neocuproine on relaxations to EFS without affecting the relaxations to NO, catalase may protect the nitrergic neurotransmitter mainly at a prejunctional site whereas Cu/ZnSOD protects at a postjunctional site.
Figures

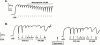
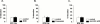
Similar articles
-
S-nitrosothiols and the nitrergic neurotransmitter in the rat gastric fundus: effect of antioxidants and metal chelation.Br J Pharmacol. 1998 Mar;123(6):1039-46. doi: 10.1038/sj.bjp.0701692. Br J Pharmacol. 1998. PMID: 9559884 Free PMC article.
-
Neocuproine potentiates the activity of the nitrergic neurotransmitter but inhibits that of S-nitrosothiols.Eur J Pharmacol. 1999 Sep 24;381(2-3):151-9. doi: 10.1016/s0014-2999(99)00564-6. Eur J Pharmacol. 1999. PMID: 10554883
-
Role of superoxide dismutase enzymes and ascorbate in protection of nitrergic relaxation against superoxide anions in mouse duodenum.Acta Pharmacol Sin. 2008 Jun;29(6):687-97. doi: 10.1111/j.1745-7254.2008.00802.x. Acta Pharmacol Sin. 2008. PMID: 18501115
-
Role of antioxidants in the protection of the nitrergic neurotransmitter.Acta Neurol Belg. 2002 Jun;102(2):68-72. Acta Neurol Belg. 2002. PMID: 12161902 Review.
-
The nitrergic transmitter of the anococcygeus--NO or not?Arch Int Pharmacodyn Ther. 1995 Jan-Feb;329(1):39-51. Arch Int Pharmacodyn Ther. 1995. PMID: 7543747 Review.
Cited by
-
Study on the cyclic GMP-dependency of relaxations to endogenous and exogenous nitric oxide in the mouse gastrointestinal tract.Br J Pharmacol. 2007 Jan;150(1):88-96. doi: 10.1038/sj.bjp.0706964. Epub 2006 Nov 20. Br J Pharmacol. 2007. PMID: 17115067 Free PMC article.
-
Biological activities of polypyridyl-type ligands: implications for bioinorganic chemistry and light-activated metal complexes.Curr Opin Chem Biol. 2021 Apr;61:191-202. doi: 10.1016/j.cbpa.2021.01.016. Epub 2021 Mar 30. Curr Opin Chem Biol. 2021. PMID: 33799087 Free PMC article. Review.
-
Functional evidence that ATP or a related purine is an inhibitory NANC neurotransmitter in the mouse jejunum: study on the identity of P2X and P2Y purinoceptors involved.Br J Pharmacol. 2003 Nov;140(6):1108-16. doi: 10.1038/sj.bjp.0705536. Epub 2003 Oct 6. Br J Pharmacol. 2003. PMID: 14530212 Free PMC article.
References
-
- BOECKXSTAENS G.E., PELCKMANS P.A., BOGERS J.J., BULT H., DE MAN J.G., OOSTERBOSCH L., HERMAN A.G., VAN MAERCKE Y.M. Release of nitric oxide upon stimulation of nonadrenergic noncholinergic nerves in the rat gastric fundus. J. Pharmacol. Exp. Ther. 1991;256:441–447. - PubMed
-
- BOECKXSTAENS G.E., PELCKMANS P.A., BULT H., DE MAN J.G., HERMAN A.G., VAN MAERCKE Y.M. Non-adrenergic non-cholinergic relaxation mediated by nitric oxide in the canine ileocolonic junction. Eur. J. Pharmacol. 1990;190:239–246. - PubMed
-
- BOECKXSTAENS G.E., PELCKMANS P.A., DE MAN J.G., BULT H., HERMAN A.G., VAN MAERCKE Y.M. Evidence for a differential release of nitric oxide and vasoactive intestinal polypeptide by nonadrenergic noncholinergic nerves in the rat gastric fundus. Arch. Int. Pharmacodyn. Ther. 1992;318:107–115. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

