Interleukin 12 p40 production by barrier epithelial cells during airway inflammation
- PMID: 11157054
- PMCID: PMC2195918
- DOI: 10.1084/jem.193.3.339
Interleukin 12 p40 production by barrier epithelial cells during airway inflammation
Abstract
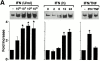
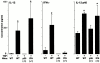
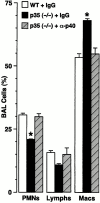
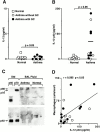
Human airway epithelial cells appear specially programmed for expression of immune response genes implicated in immunity and inflammation. To better determine how this epithelial system operates in vivo, we analyzed its behavior in mouse models that allow for in vitro versus in vivo comparison and genetic modification. Initial comparisons indicated that tumor necrosis factor alpha induction of epithelial intercellular adhesion molecule 1 required sequential induction of interleukin (IL)-12 (p70) and interferon gamma, and unexpectedly localized IL-12 production to airway epithelial cells. Epithelial IL-12 was also inducible during paramyxoviral bronchitis, but in this case, initial IL-12 p70 expression was followed by 75-fold greater expression of IL-12 p40 (as monomer and homodimer). Induction of IL-12 p40 was even further increased in IL-12 p35-deficient mice, and in this case, was associated with increased mortality and epithelial macrophage accumulation. The results placed epithelial cell overgeneration of IL-12 p40 as a key intermediate for virus-inducible inflammation and a candidate for epithelial immune response genes that are abnormally programmed in inflammatory disease. This possibility was further supported when we observed IL-12 p40 overexpression selectively in airway epithelial cells in subjects with asthma and concomitant increases in airway levels of IL-12 p40 (as homodimer) and airway macrophages. Taken together, these results suggest a novel role for epithelial-derived IL-12 p40 in modifying the level of airway inflammation during mucosal defense and disease.
Figures









References
-
- Medzhitov R., Janeway C.A., Jr. An ancient system of host defense. Curr. Opin. Immunol. 1998;10:12–15. - PubMed
-
- Holtzman M.J., Zhang V., Hussain H., Roswit W.T., Wilson J.D. Prostaglandin H synthase and lipoxygenase gene families in the epithelial cell barrier. In: Goetzl E.J., Lewis R.A., Rola-Pleszczynski M., editors. Cellular Generation, Transport and Effects of EicosanoidsBiological Roles and Pharmacological Intervention. Ann. NY. Acad. Sci; New York: 1995. pp. 58–77. - PubMed
-
- Holtzman M.J., Castro M., Look D.C., O'Sullivan M., Walter M.J. Regulation of epithelial-leukocyte interaction and epithelial immune-response genes. In: Busse W., Holgate S., editors. Asthma and Rhinitis. Blackwell Scientific; Cambridge, MA: 2000. pp. 784–800.
-
- Look D.C., Keller B.T., Rapp S.R., Holtzman M.J. Selective induction of intercellular adhesion molecule-1 by interferon-γ in human airway epithelial cells. Am. J. Physiol. 1992;263:L79–L87. - PubMed
-
- Nakajima S., Look D.C., Roswit W.T., Bragdon M.J., Holtzman M.J. Selective differences in vascular endothelial- vs. airway epithelial-T cell adhesion mechanisms. Am. J. Physiol. 1994;267:L422–L432. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

