Presenilin-1 mutations reduce cytoskeletal association, deregulate neurite growth, and potentiate neuronal dystrophy and tau phosphorylation
- PMID: 11157069
- PMCID: PMC6762317
- DOI: 10.1523/JNEUROSCI.21-03-00834.2001
Presenilin-1 mutations reduce cytoskeletal association, deregulate neurite growth, and potentiate neuronal dystrophy and tau phosphorylation
Abstract
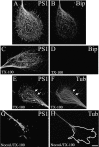
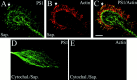
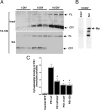
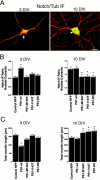
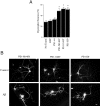
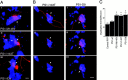
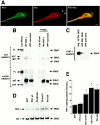
Mutations in presenilin genes are linked to early onset familial Alzheimer's disease (FAD). Previous work in non-neuronal cells indicates that presenilin-1 (PS1) associates with cytoskeletal elements and that it facilitates Notch1 signaling. Because Notch1 participates in the control of neurite growth, cultured hippocampal neurons were used to investigate the cytoskeletal association of PS1 and its potential role during neuronal development. We found that PS1 associates with microtubules (MT) and microfilaments (MF) and that its cytoskeletal association increases dramatically during neuronal development. PS1 was detected associated with MT in the central region of neuronal growth cones and with MF in MF-rich areas extending into filopodia and lamellipodia. In differentiated neurons, PS1 mutations reduced the interaction of PS1 with cytoskeletal elements, diminished the nuclear translocation of the Notch1 intracellular domain (NICD), and promoted a marked increase in total neurite length. In developing neurons, PS1 overexpression increased the nuclear translocation of NICD and inhibited neurite growth, whereas PS1 mutations M146V, I143T, and deletion of exon 9 (D9) did not facilitate NICD nuclear translocation and had no effect on neurite growth. In cultures that were treated with amyloid beta (Abeta), PS1 mutations significantly increased neuritic dystrophy and AD-like changes in tau such as hyperphosphorylation, release from MT, and increased tau protein levels. We conclude that PS1 participates in the regulation of neurite growth and stabilization in both developing and differentiated neurons. In the Alzheimer's brain PS1 mutations may promote neuritic dystrophy and tangle formation by interfering with Notch1 signaling and enhancing pathological changes in tau.
Figures
Similar articles
-
Characterisation of cytoskeletal abnormalities in mice transgenic for wild-type human tau and familial Alzheimer's disease mutants of APP and presenilin-1.Neurobiol Dis. 2004 Feb;15(1):47-60. doi: 10.1016/j.nbd.2003.09.007. Neurobiol Dis. 2004. PMID: 14751770
-
Cytoskeletal alterations differentiate presenilin-1 and sporadic Alzheimer's disease.Acta Neuropathol. 2009 Jan;117(1):19-29. doi: 10.1007/s00401-008-0458-z. Epub 2008 Nov 18. Acta Neuropathol. 2009. PMID: 19015863
-
Formation of tau inclusions in knock-in mice with familial Alzheimer disease (FAD) mutation of presenilin 1 (PS1).J Biol Chem. 2006 Feb 24;281(8):5037-41. doi: 10.1074/jbc.M509145200. Epub 2005 Dec 23. J Biol Chem. 2006. PMID: 16377636
-
Aβ Influences Cytoskeletal Signaling Cascades with Consequences to Alzheimer's Disease.Mol Neurobiol. 2015 Dec;52(3):1391-1407. doi: 10.1007/s12035-014-8913-4. Epub 2014 Oct 26. Mol Neurobiol. 2015. PMID: 25344315 Review.
-
[Understanding of molecular pathogenesis of Alzheimer's disease: implications for drug development].Nihon Yakurigaku Zasshi. 2000 Feb;115(2):79-88. doi: 10.1254/fpj.115.79. Nihon Yakurigaku Zasshi. 2000. PMID: 10876794 Review. Japanese.
Cited by
-
Diosmin reduces cerebral Aβ levels, tau hyperphosphorylation, neuroinflammation, and cognitive impairment in the 3xTg-AD mice.J Neuroimmunol. 2016 Oct 15;299:98-106. doi: 10.1016/j.jneuroim.2016.08.018. Epub 2016 Sep 1. J Neuroimmunol. 2016. PMID: 27725131 Free PMC article.
-
Presenilin/γ-secretase-dependent EphA3 processing mediates axon elongation through non-muscle myosin IIA.Elife. 2019 Oct 2;8:e43646. doi: 10.7554/eLife.43646. Elife. 2019. PMID: 31577226 Free PMC article.
-
Complex environment experience rescues impaired neurogenesis, enhances synaptic plasticity, and attenuates neuropathology in familial Alzheimer's disease-linked APPswe/PS1DeltaE9 mice.FASEB J. 2010 Jun;24(6):1667-81. doi: 10.1096/fj.09-136945. Epub 2010 Jan 19. FASEB J. 2010. PMID: 20086049 Free PMC article.
-
Aberrant accumulation of age- and disease-associated factors following neural probe implantation in a mouse model of Alzheimer's disease.J Neural Eng. 2023 Sep 1;20(4):046044. doi: 10.1088/1741-2552/aceca5. J Neural Eng. 2023. PMID: 37531953 Free PMC article.
-
Presenilin PS1∆E9 disrupts mobility of secretory organelles in rat astrocytes.Acta Physiol (Oxf). 2018 Jun;223(2):e13046. doi: 10.1111/apha.13046. Epub 2018 Feb 19. Acta Physiol (Oxf). 2018. PMID: 29392878 Free PMC article.
References
-
- Artavanis-Tsakonas S, Matsuno K, Fortini ME. Notch signaling. Science. 1995;268:225–232. - PubMed
-
- Begley JG, Duan W, Chan S, Duff K, Mattson MP. Altered calcium homeostasis and mitochondrial dysfunction in cortical synaptic compartments of presenilin-1 mutant mice. J Neurochem. 1999;72:1030–1039. - PubMed
-
- Beher D, Elle C, Underwood J, Davis JB, Ward R, Karran E, Masters CL, Beyreuther K, Multhaup G. Proteolytic fragments of Alzheimer's disease-associated presenilin-1 are present in synaptic organelles and growth cone membranes of rat brain. J Neurochem. 1999;72:1564–1573. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
