Deleted in colorectal cancer (DCC) regulates the migration of luteinizing hormone-releasing hormone neurons to the basal forebrain
- PMID: 11157077
- PMCID: PMC6762310
- DOI: 10.1523/JNEUROSCI.21-03-00911.2001
Deleted in colorectal cancer (DCC) regulates the migration of luteinizing hormone-releasing hormone neurons to the basal forebrain
Abstract
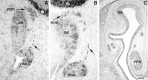
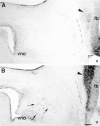
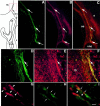
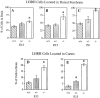
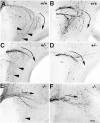
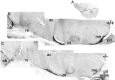
Luteinizing hormone-releasing hormone (LHRH) neurons migrate from the vomeronasal organ (VNO) to the forebrain in all mammals studied. In mice, most LHRH neuron migration is dependent on axons that originate in the VNO but bypass the olfactory bulb and project into the basal forebrain. Thus, cues that regulate the trajectories of these vomeronasal axons are candidates for determining the destination of LHRH neurons. Using in situ hybridization techniques, we examined the expression of Deleted in colorectal cancer (DCC), a vertebrate receptor for the guidance molecule netrin-1, during development of the olfactory system. DCC is expressed by cells in the olfactory epithelium (OE) and VNO, and in cells migrating from the OE and VNO from embryonic day 11 (E11) to E14. Some DCC(+) cells on vomeronasal axons in the nose also express LHRH. However, DCC expression is downregulated beginning at E12, so few if any LHRH neurons in the forebrain also express DCC. In rat, DCC is expressed on TAG-1(+) axons that guide migrating LHRH neurons. We therefore examined LHRH neuron migration in DCC(-/-) mice and found that trajectories of the caudal vomeronasal nerve and positions of LHRH neurons are abnormal. Fewer than the normal number of LHRH neurons are found in the basal forebrain, and many LHRH neurons are displaced into the cerebral cortex of DCC(-/-) mice. These results are consistent with the idea that DCC regulates the trajectories of a subset of vomeronasal axons that guide the migration of LHRH neurons. Loss of DCC function results in the migration of many LHRH neurons to inappropriate destinations.
Figures

Similar articles
-
Netrin 1-mediated chemoattraction regulates the migratory pathway of LHRH neurons.Eur J Neurosci. 2004 Jan;19(1):11-20. doi: 10.1111/j.1460-9568.2004.03094.x. Eur J Neurosci. 2004. PMID: 14750959
-
Lactosamine modulates the rate of migration of GnRH neurons during mouse development.Eur J Neurosci. 2006 Aug;24(3):654-60. doi: 10.1111/j.1460-9568.2006.04955.x. Eur J Neurosci. 2006. PMID: 16930397
-
The migration of luteinizing hormone-releasing hormone neurons in the developing rat is associated with a transient, caudal projection of the vomeronasal nerve.J Neurosci. 1995 Dec;15(12):7769-77. doi: 10.1523/JNEUROSCI.15-12-07769.1995. J Neurosci. 1995. PMID: 8613718 Free PMC article.
-
Origin and migration of luteinizing hormone-releasing hormone neurons in mammals.Microsc Res Tech. 1999 Jan 1;44(1):2-10. doi: 10.1002/(SICI)1097-0029(19990101)44:1<2::AID-JEMT2>3.0.CO;2-4. Microsc Res Tech. 1999. PMID: 9915559 Review.
-
Luteinizing hormone-releasing hormone (LHRH) and neural cell adhesion molecule (NCAM)-immunoreactivity in development of the forebrain and reproductive system.Ann Endocrinol (Paris). 1994;55(6):235-41. Ann Endocrinol (Paris). 1994. PMID: 7864579 Review.
Cited by
-
From nose to brain: development of gonadotrophin-releasing hormone-1 neurones.J Neuroendocrinol. 2010 Jul;22(7):743-53. doi: 10.1111/j.1365-2826.2010.02034.x. J Neuroendocrinol. 2010. PMID: 20646175 Free PMC article. Review.
-
Gonadotropin-releasing hormone (GnRH) neuron migration: initiation, maintenance and cessation as critical steps to ensure normal reproductive function.Front Neuroendocrinol. 2011 Jan;32(1):43-52. doi: 10.1016/j.yfrne.2010.07.005. Epub 2010 Jul 30. Front Neuroendocrinol. 2011. PMID: 20650288 Free PMC article. Review.
-
The Differential Roles for Neurodevelopmental and Neuroendocrine Genes in Shaping GnRH Neuron Physiology and Deficiency.Int J Mol Sci. 2021 Aug 30;22(17):9425. doi: 10.3390/ijms22179425. Int J Mol Sci. 2021. PMID: 34502334 Free PMC article. Review.
-
Slit2 and Robo3 modulate the migration of GnRH-secreting neurons.Development. 2012 Sep;139(18):3326-31. doi: 10.1242/dev.079418. Development. 2012. PMID: 22912413 Free PMC article.
-
Molecular clues in the regulation of mini-puberty involve neuronal DNA binding transcription factor NHLH2.Basic Clin Androl. 2021 Mar 18;31(1):6. doi: 10.1186/s12610-021-00124-w. Basic Clin Androl. 2021. PMID: 33731013 Free PMC article. Review.
References
-
- Ackerman SL, Kozak LP, Przyborski SA, Rund LA, Boyer BB, Knowles BB. The mouse rostral cerebellar malformation gene encodes an UNC-5-like protein. Nature. 1997;286:838–842. - PubMed
-
- Bless EP, Westaway WA, Schwarting GA, Tobet SA. Effects of gamma-aminobutyric acid (A) receptor manipulation on migrating gonadotropin-releasing hormone neurons through the entire migratory route in vivo and in vitro. Endocrinology. 2000a;141:1254–1262. - PubMed
-
- Bless EP, Pays L, Schwarting GA, Tobet SA. Expression of a cell surface glycoconjugate in gonadotropin releasing hormone (GnRH) neurons in mice. Soc Neurosci Abstr. 2000b;25:1350.
-
- Brown A, Yates PA, Burrola P, Ortuno D, Vaidya A, Jessell TM, Pfaff SL, O'Leary DDM, Lemke G. Topographic mapping from the retina to the midbrain is controlled by relative but not absolute levels of EphA receptor signaling. Cell. 2000;102:77–88. - PubMed
-
- Colamarino SA, Tessier-Lavigne M. The axonal chemoattractant netrin-1 is also a chemorepellent for trochlear motor axons. Cell. 1995;81:187–195. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
