DIABLO promotes apoptosis by removing MIHA/XIAP from processed caspase 9
- PMID: 11157976
- PMCID: PMC2195997
- DOI: 10.1083/jcb.152.3.483
DIABLO promotes apoptosis by removing MIHA/XIAP from processed caspase 9
Abstract
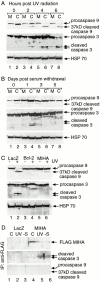
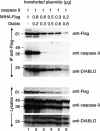
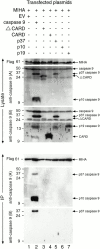
MIHA is an inhibitor of apoptosis protein (IAP) that can inhibit cell death by direct interaction with caspases, the effector proteases of apoptosis. DIABLO is a mammalian protein that can bind to IAPs and antagonize their antiapoptotic effect, a function analogous to that of the proapoptotic Drosophila molecules, Grim, Reaper, and HID. Here, we show that after UV radiation, MIHA prevented apoptosis by inhibiting caspase 9 and caspase 3 activation. Unlike Bcl-2, MIHA functioned after release of cytochrome c and DIABLO from the mitochondria and was able to bind to both processed caspase 9 and processed caspase 3 to prevent feedback activation of their zymogen forms. Once released into the cytosol, DIABLO bound to MIHA and disrupted its association with processed caspase 9, thereby allowing caspase 9 to activate caspase 3, resulting in apoptosis.
Figures


References
-
- Day C.L., Dupont C., Lackmann M., Vaux D.L., Hinds M.G. Solution structure and mutagenesis of the caspase recruitment domain (CARD) from Apaf-1. Cell Death Differ. 1999;6:1125–1132. - PubMed
-
- Deveraux Q.L., Takahashi R., Salvesen G.S., Reed J.C. X-linked IAP is a direct inhibitor of cell-death proteases. Nature. 1997;388:300–304. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

