Analysis of growth characteristics of filamentous fungi in different nutrient media
- PMID: 11158093
- PMCID: PMC87762
- DOI: 10.1128/JCM.39.2.478-484.2001
Analysis of growth characteristics of filamentous fungi in different nutrient media
Abstract
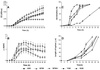
A microbroth kinetic model based on turbidity measurements was developed in order to analyze the growth characteristics of three species of filamentous fungi (Rhizopus microsporus, Aspergillus fumigatus, and Scedosporium prolificans) characterized by different growth rates in five nutrient media (antibiotic medium 3, yeast nitrogen base medium, Sabouraud broth, RPMI 1640 alone, and RPMI 1640 with 2% glucose). In general, five distinct phases in the growth of filamentous fungi could be distinguished, namely, the lag phase, the first transition period, the log phase, the second transition period, and the stationary phase. The growth curves were smooth and were characterized by the presence of long transition periods. Among the different growth phases distinguished, the smallest variability in growth rates among the strains of each species was found during the log phase in all nutrient media. The different growth phases of filamentous fungi were barely distinguishable in RPMI 1640, in which the poorest growth was observed for all fungi even when the medium was supplemented with 2% glucose. R. microsporus and A. fumigatus grew better in Sabouraud and yeast nitrogen base medium than in RPMI 1640, with growth rates three to four times higher. None of the media provided optimal growth of S. prolificans. The germination of Rhizopus spores and Aspergillus and Scedosporium conidia commenced after 2 and 5 h of incubation, respectively. The elongation rates ranged from 39.6 to 26.7, 25.4 to 20.2, and 16.9 to 9.9 microm/h for Rhizopus, Aspergillus, and Scedoporium hyphae, respectively. The germination of conidia and spores and the elongation rates of hyphae were enhanced in antibiotic medium 3 and delayed in yeast nitrogen base medium. In conclusion, the growth curves provide a useful tool to gain insight into the growth characteristics of filamentous fungi in different nutrient media and may help to optimize the methodology for antifungal susceptibility testing.
Figures


Similar articles
-
Conidial germination in Scedosporium apiospermum, S. aurantiacum, S. minutisporum and Lomentospora prolificans: influence of growth conditions and antifungal susceptibility profiles.Mem Inst Oswaldo Cruz. 2016 Jun 27;0(7):0. doi: 10.1590/0074-02760160200. Mem Inst Oswaldo Cruz. 2016. PMID: 27355215 Free PMC article.
-
[Identification of filamentous fungi isolated from clinical samples by two different methods and their susceptibility results].Mikrobiyol Bul. 2012 Jan;46(1):65-78. Mikrobiyol Bul. 2012. PMID: 22399173 Turkish.
-
In vitro antifungal susceptibility testing of filamentous fungi with Sensititre Yeast One.Mycoses. 2006 Jul;49(4):293-7. doi: 10.1111/j.1439-0507.2006.01250.x. Mycoses. 2006. PMID: 16784443
-
Use of turbidimetric growth curves for early determination of antifungal drug resistance of filamentous fungi.J Clin Microbiol. 2003 Oct;41(10):4718-25. doi: 10.1128/JCM.41.10.4718-4725.2003. J Clin Microbiol. 2003. PMID: 14532210 Free PMC article.
-
Pulmonary defense mechanisms against opportunistic fungal pathogens.Immunol Ser. 1989;47:243-71. Immunol Ser. 1989. PMID: 2490078 Review.
Cited by
-
Redefining the Subsurface Biosphere: Characterization of Fungi Isolated From Energy-Limited Marine Deep Subsurface Sediment.Front Fungal Biol. 2021 Sep 24;2:727543. doi: 10.3389/ffunb.2021.727543. eCollection 2021. Front Fungal Biol. 2021. PMID: 37744089 Free PMC article.
-
Conidial germination in Scedosporium apiospermum, S. aurantiacum, S. minutisporum and Lomentospora prolificans: influence of growth conditions and antifungal susceptibility profiles.Mem Inst Oswaldo Cruz. 2016 Jun 27;0(7):0. doi: 10.1590/0074-02760160200. Mem Inst Oswaldo Cruz. 2016. PMID: 27355215 Free PMC article.
-
Human Neutrophils Are Primed by Chemoattractant Gradients for Blocking the Growth of Aspergillus fumigatus.J Infect Dis. 2016 Feb 1;213(3):465-75. doi: 10.1093/infdis/jiv419. Epub 2015 Aug 12. J Infect Dis. 2016. PMID: 26272935 Free PMC article.
-
Pentamidine is active in vitro against Fusarium species.Antimicrob Agents Chemother. 2003 Oct;47(10):3252-9. doi: 10.1128/AAC.47.10.3252-3259.2003. Antimicrob Agents Chemother. 2003. PMID: 14506038 Free PMC article.
-
Utilisation of carbon substrates by orchid and ericoid mycorrhizal fungi from Australian dry sclerophyll forests.Mycorrhiza. 2006 May;16(3):175-182. doi: 10.1007/s00572-005-0029-2. Epub 2005 Dec 22. Mycorrhiza. 2006. PMID: 16374622
References
-
- Cormican M G, Pfaller M A. Standardization of antifungal susceptibility testing. J Antimicrob Chemother. 1996;38:561–578. - PubMed
-
- Espinel-Ingroff A, Barchiesi F, Hazen K C, Martinez-Suarez J V, Scalise G. Standardization of antifungal susceptibility testing and clinical relevance. Med Mycol. 1998;36(Suppl. 1):68–78. - PubMed
-
- Espinel-Ingroff A, Bartlett M, Bowden R, Chin N X, Cooper C, Jr, Fothergill A, McGinnis M R, Menezes P, Messer S A, Nelson P W, Odds F C, Pasarell L, Peter J, Pfaller M A, Rex J H, Rinaldi M G, Shankland G S, Walsh T J, Weitzman I. Multicenter evaluation of proposed standardized procedure for antifungal susceptibility testing of filamentous fungi. J Clin Microbiol. 1997;35:139–143. - PMC - PubMed
-
- Espinel-Ingroff A, Dawson K, Pfaller M, Anaissie E, Breslin B, Dixon D, Fothergill A, Paetznick V, Peter J, Rinaldi M G, Walsh T. Comparative and collaborative evaluation of standardization of antifungal susceptibility testing for filamentous fungi. Antimicrob Agents Chemother. 1995;39:314–319. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

