Age-dependent induction of congophilic neurofibrillary tau inclusions in tau transgenic mice
- PMID: 11159192
- PMCID: PMC1850303
- DOI: 10.1016/S0002-9440(10)63997-1
Age-dependent induction of congophilic neurofibrillary tau inclusions in tau transgenic mice
Abstract
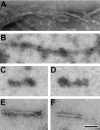
Intraneuronal filamentous tau inclusions such as neurofibrillary tangles (NFTs) are neuropathological hallmarks of Alzheimer's disease (AD) and related sporadic and familial tauopathies. NFTs identical to those found in AD brains have also been detected in the hippocampus and entorhinal cortex of cognitively normal individuals as they age. To recapitulate age-induced NFT formation in a mouse model, we examined 12- to 24-month-old transgenic (Tg) mice overexpressing the smallest human brain tau isoform. These Tg mice develop congophilic tau inclusions in several brain regions including the hippocampus, amygdala, and entorhinal cortex. NFT-like inclusions were first detected in Tg mice at 18 to 20 months of age and they were detected by histochemical dyes that bind specifically to crossed beta-pleated sheet structures (eg, Congo red, Thioflavin S). Moreover, ultrastructurally these lesions contained straight tau filaments comprised of both mouse and human tau proteins but not other cytoskeletal proteins (eg, neurofilaments, microtubules). Isolated tau filaments were also recovered from detergent-insoluble tau fractions and insoluble tau proteins accumulated in brain in an age-dependent manner. Thus, overexpression of the smallest human brain tau isoform resulted in late onset and age-dependent formation of congophilic tau inclusions with properties similar to those in the tangles of human tauopathies, thereby implicating aging in the pathogenesis of fibrous tau inclusions.
Figures
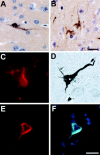
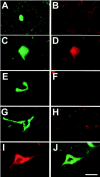
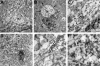
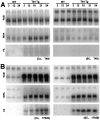
References
-
- Hong M, Trojanowski JQ, Lee VMY: Tau-based neurofibrillary lesions. Clark CM Trojanowski JQ eds. Neurodegenerative Dementias: Clinical Features and Pathological Mechanisms. 2000, :pp 161-176 McGraw Hill, New York
-
- Lee VMY, Trojanowski JQ: Distinct tau gene mutations induce specific dysfunctions/toxic properties in tau proteins associated with specific FTDP-17 phenotypes. Lee VMY Trojanowski JQ Buee L Christen Y eds. Fatal Attractions: Protein Aggregates in Neurodegenerative Diseases. 2000, :pp 87-104 IPSEN Foundation, Paris
-
- Hof PR, Bouras C, Morrison JH: Cortical Neuropathology In Aging And Dementing Disorders: Neuronal Typology, Connectivity And Selective Vulnerability. Peters A Morrison JH eds. Cerebral Cortex 14. 1999, :pp 175-276 Kluwer Academic/Plenum Publishers, New York
-
- Andreadis A, Brown WM, Kosik KS: Structure and novel exons of the human tau gene. Biochemistry 1992, 31:10626-10633 - PubMed
-
- Goedert M, Spillantini MG, Jakes R, Rutherford D, Crowther RA: Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer’s disease. Neuron 1989, 3:519-526 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

