A disease-associated cellular immune response in type 1 diabetics to an immunodominant epitope of insulin
- PMID: 11160133
- PMCID: PMC198872
- DOI: 10.1172/JCI8525
A disease-associated cellular immune response in type 1 diabetics to an immunodominant epitope of insulin
Abstract
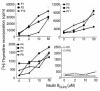
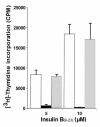
The 9-23 amino acid region of the insulin B chain (B9-23) is a dominant epitope recognized by pathogenic T lymphocytes in nonobese diabetic mice, the animal model for type 1 diabetes. We describe herein similar (B9-23)-specific T-cell responses in peripheral lymphocytes obtained from patients with recent-onset type 1 diabetes and from prediabetic subjects at high risk for disease. Short-term T-cell lines generated from patient peripheral lymphocytes showed significant proliferative responses to (B9-23), whereas lymphocytes isolated from HLA and/or age-matched nondiabetic normal controls were unresponsive. Antibody-mediated blockade demonstrated that the response was HLA class II restricted. Use of the highly sensitive cytokine-detection ELISPOT assay revealed that these (B9-23)-specific cells were present in freshly isolated lymphocytes from only the type 1 diabetics and prediabetics and produced the proinflammatory cytokine IFN-gamma. This study is, to our knowledge, the first demonstration of a cellular response to the (B9-23) insulin epitope in human type 1 diabetes and suggests that the mouse and human diseases have strikingly similar autoantigenic targets, a feature that should facilitate development of antigen-based therapeutics.
Figures

References
-
- Atkinson MA, Maclaren NK. The pathogenesis of insulin-dependent diabetes mellitus. N Engl J Med. 1994;331:1428–1436. - PubMed
-
- Delovitch TL, Singh B. The nonobese diabetic mouse as a model of autoimmune diabetes: immune dysregulation gets the NOD [erratum 1998, 8:531] Immunity. 1997;7:727–738. - PubMed
-
- Durinovic-Bello I. Autoimmune diabetes: the role of T cells, MHC molecules and autoantigens. Autoimmunity. 1998;27:159–177. - PubMed
-
- Wegmann DR, Norbury-Glaser M, Daniel D. Insulin-specific T cells are a predominant component of islet infiltrates in pre-diabetic NOD mice. Eur J Immunol. 1994;24:1853–1857. - PubMed
-
- Daniel D, Gill RG, Schloot N, Wegmann D. Epitope specificity, cytokine production profile and diabetogenic activity of insulin-specific T cell clones isolated from NOD mice. Eur J Immunol. 1995;25:1056–1062. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

