A new model for nuclear envelope breakdown
- PMID: 11179431
- PMCID: PMC30959
- DOI: 10.1091/mbc.12.2.503
A new model for nuclear envelope breakdown
Abstract
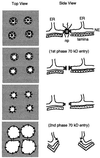
Nuclear envelope breakdown was investigated during meiotic maturation of starfish oocytes. Fluorescent 70-kDa dextran entry, as monitored by confocal microscopy, consists of two phases, a slow uniform increase and then a massive wave. From quantitative analysis of the first phase of dextran entry, and from imaging of green fluorescent protein chimeras, we conclude that nuclear pore disassembly begins several minutes before nuclear envelope breakdown. The best fit for the second phase of entry is with a spreading disruption of the membrane permeability barrier determined by three-dimensional computer simulations of diffusion. We propose a new model for the mechanism of nuclear envelope breakdown in which disassembly of the nuclear pores leads to a fenestration of the nuclear envelope double membrane.
Figures








References
-
- Collas P. Sequential PKC-, and Cdc2-mediated, phosphorylation events elicit zebrafish nuclear envelope disassembly. J Cell Sci. 1999;112:977–987. - PubMed
-
- Favreau C, Worman HJ, Wozniak RW, Frappier T, Courvaline J-C. Cell cycle-dependent phosphorylation of nucleoporins and nuclear pore membrane protein gfp210. Biochemistry. 1996;35:8035–8044. - PubMed
-
- Gerace L, Blobel G. The nuclear envelope lamina is reversibly depolymerized during mitosis. Cell. 1980;19:277–287. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

