Developments toward diagnostic breast cancer imaging using near-infrared optical measurements and fluorescent contrast agents
- PMID: 11191107
- PMCID: PMC1507982
- DOI: 10.1038/sj.neo.7900118
Developments toward diagnostic breast cancer imaging using near-infrared optical measurements and fluorescent contrast agents
Abstract
The use of near-infrared (NIR) light to interrogate deep tissues has enormous potential for molecular-based imaging when coupled with NIR excitable dyes. More than a decade has now passed since the initial proposals for NIR optical tomography for breast cancer screening using time-dependent measurements of light propagation in the breast. Much accomplishment in the development of optical mammography has been demonstrated, most recently in the application of time-domain, frequency-domain, and continuous-wave measurements that depend on endogenous contrast owing to angiogenesis and increased hemoglobin absorbance for contrast. Although exciting and promising, the necessity of angiogenesis-mediated absorption contrast for diagnostic optical mammography minimizes the potential for using NIR techniques to assess sentinel lymph node staging, metastatic spread, and multifocality of breast disease, among other applications. In this review, we summarize the progress made in the development of optical mammography, and focus on the emerging work underway in the use of diagnostic contrast agents for the molecular-based, diagnostic imaging of breast.
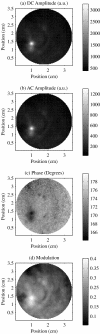
Figures
















Similar articles
-
Fluorescence-enhanced, near infrared diagnostic imaging with contrast agents.Curr Opin Chem Biol. 2002 Oct;6(5):642-50. doi: 10.1016/s1367-5931(02)00356-3. Curr Opin Chem Biol. 2002. PMID: 12413549 Review.
-
Near-infrared fluorescent dyes for enhanced contrast in optical mammography: phantom experiments.J Biomed Opt. 2001 Apr;6(2):134-40. doi: 10.1117/1.1350561. J Biomed Opt. 2001. PMID: 11375722
-
Development of high-sensitivity near-infrared fluorescence imaging device for early cancer detection.Biomed Instrum Technol. 2005 Jan-Feb;39(1):75-85. doi: 10.2345/0899-8205(2005)39[75:DOHNFI]2.0.CO;2. Biomed Instrum Technol. 2005. PMID: 15742853
-
Three-dimensional diffuse optical tomography in the parallel plane transmission geometry: evaluation of a hybrid frequency domain/continuous wave clinical system for breast imaging.Med Phys. 2003 Feb;30(2):235-47. doi: 10.1118/1.1534109. Med Phys. 2003. PMID: 12607841 Clinical Trial.
-
[Contrast media for optical mammography].Radiologe. 1997 Sep;37(9):749-55. doi: 10.1007/s001170050277. Radiologe. 1997. PMID: 9424621 Review. German.
Cited by
-
Molecular imaging of experimental abdominal aortic aneurysms.ScientificWorldJournal. 2013 Apr 23;2013:973150. doi: 10.1155/2013/973150. Print 2013. ScientificWorldJournal. 2013. PMID: 23737735 Free PMC article. Review.
-
Optical imaging of kidney cancer with novel near infrared heptamethine carbocyanine fluorescent dyes.J Urol. 2013 Feb;189(2):702-710. doi: 10.1016/j.juro.2012.09.056. Epub 2012 Sep 20. J Urol. 2013. PMID: 23000848 Free PMC article.
-
Spectral distortion in diffuse molecular luminescence tomography in turbid media.J Appl Phys. 2009 May 15;105(10):102024. doi: 10.1063/1.3116130. Epub 2009 May 19. J Appl Phys. 2009. PMID: 20157444 Free PMC article.
-
Tissue drug concentration determines whether fluorescence or absorption measurements are more sensitive in diffuse optical tomography of exogenous contrast agents.Appl Opt. 2009 Apr 1;48(10):D262-72. doi: 10.1364/ao.48.00d262. Appl Opt. 2009. PMID: 19340118 Free PMC article.
-
Cortical Network Response to Acupuncture and the Effect of the Hegu Point: An fNIRS Study.Sensors (Basel). 2019 Jan 18;19(2):394. doi: 10.3390/s19020394. Sensors (Basel). 2019. PMID: 30669377 Free PMC article.
References
-
- Doornbos RMP, Lang R, Aalders MC, Cross FW, Sterenborg HJCM. The determination of in vivo human tissue optical properties and absolute chromophore concentrations using spatially resolved steady-state diffuse reflectance spectroscopy. Phys Med Biol. 1999;44:967–981. - PubMed
-
- Balgi G, Reynolds JS, Mayer RH, Cooley R, Sevick-Muraca EM. Measurements of multiply scattered light for on-line monitoring of changes in size distribution of cell-debris suspensions. Biotechnol Prog. 1999;15:1106–1114. - PubMed
-
- Grosenick D, Wabnitz H, Rinnebert HH, Moesta KT, Schlag PM. Development of a time-domain optical mammography and first in vivo applications. Appl Opt. 1999;38:2927–2943. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
