Epitope focus, clonal composition and Th1 phenotype of the human CD4 response to the secretory mycobacterial antigen Ag85
- PMID: 11207652
- PMCID: PMC1905982
- DOI: 10.1046/j.1365-2249.2001.01450.x
Epitope focus, clonal composition and Th1 phenotype of the human CD4 response to the secretory mycobacterial antigen Ag85
Abstract
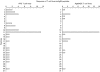
Lymphoproliferation of healthy donors was tested against mycobacterial antigens (PPD, Ag85, Ag85 peptides). All PPD responders recognized the secretory antigen Ag85 and the peptide specificity for Ag85B was defined. Peptide 91-108 was recognized by 85% of donors. In addition, all CD4 T cell lines generated from 12 donors against PPD or Ag85 responded to 91-108. When this peptide was used to generate T cell lines, the cells responded also to tuberculins from atypical mycobacterial species. Thus the cross-reactive peptide behaved as quasi-universal. The analysis of TCR-BV gene usage by cell lines showed that most Ag85-specific T cells correspond to 91-108-specific clonotypes. Intracytoplasmic staining of cell lines after phorbol myristate acetate stimulation resulted in dominance of interferon-gamma (IFN-gamma)-IL-4 double-positive cells, whereas antigen stimulation resulted in production of IFN-gamma only. The data show that peptide 91-108 is the major focus of the CD4 response to mycobacterial antigens in peripheral blood mononuclear cells and in T cell lines from PPD responders.
Figures




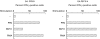
References
-
- Fine PEM. The BCG story: lessons from the past and implications for the future. Rev Infect Dis. 1989;11:S353–9. - PubMed
-
- Young DB, Kaufmann SHE, Hermans PWM, Thole JER. Mycobacterial protein antigens: a compilation. Molec Microbiol. 1992;6:133–45. - PubMed
-
- Lamb JR, Lathigra R, Rothbard JB, Sweetser D, Young RA, Ivanyi J, Young DB. Identification of mycobacterial antigens recognized by T lymphocytes. Rev Infect Dis. 1989;11:S443–7. - PubMed
-
- Horwitz MA. A new TB vaccine. Immunologist. 1997;5:15–20.
-
- Kauffmann S. Immunity to intracellular bacteria. Annu Rev Immunol. 1993;11:129–63. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

