Proton sensitivity of rat cerebellar granule cell GABAA receptors: dependence on neuronal development
- PMID: 11208970
- PMCID: PMC2278406
- DOI: 10.1111/j.1469-7793.2001.0219l.x
Proton sensitivity of rat cerebellar granule cell GABAA receptors: dependence on neuronal development
Abstract
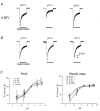
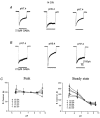
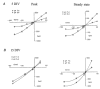
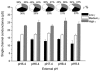
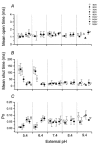
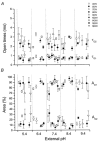
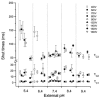
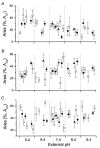
The effect of GABAA receptor development in culture on the modulation of GABA-induced currents by external H+ was examined in cerebellar granule cells using whole-cell and single-channel recording. Equilibrium concentration-response curves revealed a lower potency for GABA between 11 and 12 days in vitro (DIV) resulting in a shift of the EC50 from 10.7 to 2.4 uM. For granule cells before 11 DIV, the peak GABA-activated current was inhibited at low external pH and enhanced at high pH with a pKa of 6.6. For the steady-state response, low pH was inhibitory with a pKa of 5.56. After 11 DIV, the peak GABA-activated current was largely pH insensitive; however, the steady-state current was potentiated at low pH with a pKa of 6.84. Single GABA-activated ion channels were recorded from outside-out patches of granule cell bodies. At pH 5.4-9.4, single GABA channels exhibited multiple conductance states occurring at 22-26, 16-17 and 12-14 pS. The conductance levels were not significantly altered over the time period of study, nor by changing the external H+ concentration. Two exponential functions were required to fit the open-time frequency histograms at both early (< 11 DIV) and late (> 11 DIV) development times at each H+ concentration. The short and long open time constants were unaffected either by the extracellular H+ concentration or by neuronal development. The distribution of all shut times was fitted by the sum of three exponentials designated as short, intermediate and long. At acidic pH, the long shut time constant decreased with development as did the relative contribution of these components to the overall distribution. This was concurrent with an increase in the mean probability of channel opening. In conclusion, this study demonstrates in cerebellar granule cells that external pH can either reduce, have no effect on, or enhance GABA-activated responses depending on the stage of development, possibly related to the subunit composition of the GABAA receptors. The mode of interaction of H+ at the single-channel level and implications of such interactions at cerebellar granule cell GABAA receptors are discussed.
Figures


References
-
- Angelotti TP, Tan F, Chahine KG, Macdonald RL. Molecular and electrophysiological characterization of an allelic variant of the rat α6 GABAA receptor subunit. Molecular Brain Research. 1992;16:173–178. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

