Long-range cortical synchronization without concomitant oscillations in the somatosensory system of anesthetized cats
- PMID: 11222669
- PMCID: PMC6762928
- DOI: 10.1523/JNEUROSCI.21-05-01795.2001
Long-range cortical synchronization without concomitant oscillations in the somatosensory system of anesthetized cats
Abstract
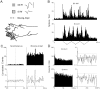
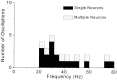
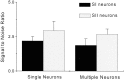
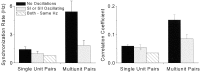
To determine whether neuronal oscillations are essential for long-range cortical synchronization in the somatosensory system, we characterized the incidence and response properties of gamma range oscillations (20-80 Hz) among pairs of synchronized neurons in primary (SI) and secondary (SII) somatosensory cortex. Synchronized SI and SII discharges, which occurred within a 3 msec period, were detected in 13% (80 of 621) of single-unit pairs and 25% (29 of 118) of multiunit pairs. Power spectra derived from the auto-correlation histograms (ACGs) revealed that approximately 15% of the neurons forming synchronized pairs were characterized by oscillations. Although 24% of the synchronized neuron pairs (19/80) were characterized by oscillations in one or both neurons, only 1% (1/80) of these pairs displayed oscillations at the same frequency in both neurons. Similar results were observed among pairs of multiunit responses. When single-trial responses were analyzed, the vast majority of responses still did not exhibit oscillations in the gamma frequency range. These results suggest that separate populations of cortical neurons can be bound together without being constrained by the phase relationships defined by specific oscillatory frequencies.
Figures





Similar articles
-
Pervasive synchronization of local neural networks in the secondary somatosensory cortex of cats during focal cutaneous stimulation.Exp Brain Res. 2002 Nov;147(2):227-42. doi: 10.1007/s00221-002-1233-3. Epub 2002 Oct 1. Exp Brain Res. 2002. PMID: 12410338
-
Cross-correlation analysis reveals laminar differences in thalamocortical interactions in the somatosensory system.J Neurophysiol. 1996 Apr;75(4):1444-57. doi: 10.1152/jn.1996.75.4.1444. J Neurophysiol. 1996. PMID: 8727389
-
Synchronization of neurons during local field potential oscillations in sensorimotor cortex of awake monkeys.J Neurophysiol. 1996 Dec;76(6):3968-82. doi: 10.1152/jn.1996.76.6.3968. J Neurophysiol. 1996. PMID: 8985893
-
Synchronization of local neural networks in the somatosensory cortex: A comparison of stationary and moving stimuli.J Neurophysiol. 1999 Mar;81(3):999-1013. doi: 10.1152/jn.1999.81.3.999. J Neurophysiol. 1999. PMID: 10085328
-
Abnormal neural oscillations and synchrony in schizophrenia.Nat Rev Neurosci. 2010 Feb;11(2):100-13. doi: 10.1038/nrn2774. Nat Rev Neurosci. 2010. PMID: 20087360 Review.
Cited by
-
Timing and connectivity in the human somatosensory cortex from single trial mass electrical activity.Hum Brain Mapp. 2002 Apr;15(4):231-46. doi: 10.1002/hbm.10023. Hum Brain Mapp. 2002. PMID: 11835611 Free PMC article.
-
Widespread spatial integration in primary somatosensory cortex.Proc Natl Acad Sci U S A. 2008 Jul 22;105(29):10233-7. doi: 10.1073/pnas.0803800105. Epub 2008 Jul 15. Proc Natl Acad Sci U S A. 2008. PMID: 18632579 Free PMC article.
-
Intercolumnar synchronization of neuronal activity in rat barrel cortex during patterned airjet stimulation: a laminar analysis.Exp Brain Res. 2006 Mar;169(3):311-25. doi: 10.1007/s00221-005-0152-5. Epub 2005 Nov 12. Exp Brain Res. 2006. PMID: 16284753
-
Effects of spatiotemporal stimulus properties on spike timing correlations in owl monkey primary somatosensory cortex.J Neurophysiol. 2012 Dec;108(12):3353-69. doi: 10.1152/jn.00414.2011. Epub 2012 Sep 26. J Neurophysiol. 2012. PMID: 23019003 Free PMC article.
-
Neural networks a century after Cajal.Brain Res Rev. 2007 Oct;55(2):264-84. doi: 10.1016/j.brainresrev.2007.06.003. Epub 2007 Jul 13. Brain Res Rev. 2007. PMID: 17692925 Free PMC article. Review.
References
-
- Aertsen AMHJ, Gerstein GL, Habib MK, Palm G. Dynamics of neuronal firing correlation: modulation of “effective connectivity.”. J Neurophysiol. 1989;61:900–917. - PubMed
-
- Ahissar M, Ahissar E, Bergman H, Vaadia E. Encoding of sound-source location and movement: activity of single neurons and interactions between adjacent neurons in the monkey auditory cortex. J Neurophysiol. 1992;67:203–215. - PubMed
-
- Alloway KD, Burton H. Homotypical ipsilateral cortical projections between somatosensory areas I and II in the cat. Neuroscience. 1985;14:15–35. - PubMed
-
- Alloway KD, Johnson MJ, Aaron GB. A comparative analysis of coordinated neuronal activity in the thalamic ventrobasal complex of rats and cats. Brain Res. 1995;691:46–56. - PubMed
-
- Alloway KD, Wallace MB, Johnson MJ. Cross-correlation analysis of cuneothalamic interactions in the rat somatosensory system: influence of receptive field topography and comparisons with thalamocortical interactions. J Neurophysiol. 1994;72:1949–1972. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
