FKBP12, the 12-kDa FK506-binding protein, is a physiologic regulator of the cell cycle
- PMID: 11226255
- PMCID: PMC30154
- DOI: 10.1073/pnas.041614198
FKBP12, the 12-kDa FK506-binding protein, is a physiologic regulator of the cell cycle
Abstract
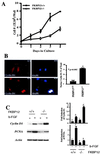
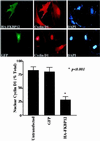
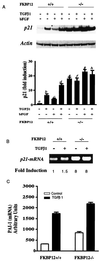
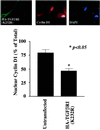
FKBP12, the 12-kDa FK506-binding protein, is a ubiquitous abundant protein that acts as a receptor for the immunosuppressant drug FK506, binds tightly to intracellular calcium release channels and to the transforming growth factor beta (TGF-beta) type I receptor. We now demonstrate that cells from FKBP12-deficient (FKBP12(-/-)) mice manifest cell cycle arrest in G(1) phase and that these cells can be rescued by FKBP12 transfection. This arrest is mediated by marked augmentation of p21(WAF1/CIP1) levels, which cannot be further augmented by TGF-beta1. The p21 up-regulation and cell cycle arrest derive from the overactivity of TGF-beta receptor signaling, which is normally inhibited by FKBP12. Cell cycle arrest is prevented by transfection with a dominant-negative TGF-beta receptor construct. TGF-beta receptor signaling to gene expression can be mediated by SMAD, p38, and ERK/MAP kinase (extracellular signal-regulated kinase/mitogen-activated protein kinase) pathways. SMAD signaling is down-regulated in FKBP12(-/-) cells. Inhibition of ERK/MAP kinase fails to affect p21 up-regulation. By contrast, activated phosphorylated p38 is markedly augmented in FKBP12(-/-) cells and the p21 up-regulation is prevented by an inhibitor of p38. Thus, FKBP12 is a physiologic regulator of cell cycle acting by normally down-regulating TGF-beta receptor signaling.
Figures


References
-
- Liu J, Farmer J D, Jr, Lane W S, Friedman J, Weissman I, Schreiber S L. Cell. 1991;66:807–815. - PubMed
-
- Friedman J, Weissman I. Cell. 1991;66:799–806. - PubMed
-
- Snyder S H, Lai M M, Burnett P E. Neuron. 1998;21:283–294. - PubMed
-
- Cameron A M, Steiner J P, Roskams A J, Ali S M, Ronnett G V, Snyder S H. Cell. 1995;83:463–472. - PubMed
-
- Brillantes A B, Ondrias K, Scott A, Kobrinsky E, Ondriasova E, Moschella M C, Jayaraman T, Landers M, Ehrlich B E, Marks A R. Cell. 1994;77:513–523. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

