Molecular characterization of cryptosporidium oocysts in samples of raw surface water and wastewater
- PMID: 11229897
- PMCID: PMC92700
- DOI: 10.1128/AEM.67.3.1097-1101.2001
Molecular characterization of cryptosporidium oocysts in samples of raw surface water and wastewater
Abstract
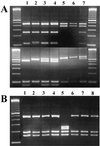
Recent molecular characterizations of Cryptosporidium parasites make it possible to differentiate the human-pathogenic Cryptosporidium parasites from those that do not infect humans and to track the source of Cryptosporidium oocyst contamination in the environment. In this study, we used a small-subunit rRNA-based PCR-restriction fragment length polymorphism (RFLP) technique to detect and characterize Cryptosporidium oocysts in 55 samples of raw surface water collected from several areas in the United States and 49 samples of raw wastewater collected from Milwaukee, Wis. Cryptosporidium parasites were detected in 25 surface water samples and 12 raw wastewater samples. C. parvum human and bovine genotypes were the dominant Cryptosporidium parasites in the surface water samples from sites where there was potential contamination by humans and cattle, whereas C. andersoni was the most common parasite in wastewater. There may be geographic differences in the distribution of Cryptosporidium genotypes in surface water. The PCR-RFLP technique can be a useful alternative method for detection and differentiation of Cryptosporidium parasites in water.
Figures
References
-
- Chung E, Aldom J E, Carreno R A, Chagla A H, Kostrzynska M, Lee H, Palmateer G, Trevors J T, Unger S, Xu R, De Grandis S A. PCR-based quantitation of Cryptosporidium parvum in municipal water samples. J Microbiol Methods. 1999;38:119–130. - PubMed
-
- Chung E, Aldom J E, Chagla A H, Kostrzynska M, Lee H, Palmateer G, Trevors J T, Unger S, Degrandis S. Detection of Cryptosporidium parvum oocysts in municipal water samples by the polymerase chain reaction. J Microbiol Methods. 1998;33:171–180.
-
- Di Giovanni G D, Hashemi F H, Shaw N J, Abrams F A, LeChevallier M W, Abbaszadegan M. Detection of infectious Cryptosporidium parvum oocysts in surface and filter backwash water samples by immunomagnetic separation and integrated cell culture-PCR. Appl Environ Microbiol. 1999;65:3427–3432. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

