The transmembrane form of the CX3CL1 chemokine fractalkine is expressed predominantly by epithelial cells in vivo
- PMID: 11238035
- PMCID: PMC1850344
- DOI: 10.1016/S0002-9440(10)64034-5
The transmembrane form of the CX3CL1 chemokine fractalkine is expressed predominantly by epithelial cells in vivo
Abstract
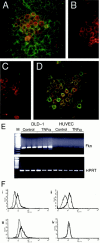
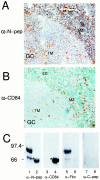
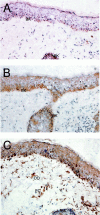
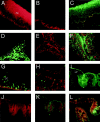
Fractalkine (CX3CL1) is synthesized as a type I transmembrane protein. Its unique CX(3)C chemokine domain is attached to a 241-amino acid mucin stalk, a 19-amino acid transmembrane domain, and a 37-amino acid intracellular domain of unknown function. A soluble form of fractalkine can be generated by proteolytic cleavage at the base of the mucin stalk. Novel monoclonal and polyclonal antibodies that specifically recognize only the amino- or carboxyl-terminal ends of the human fractalkine molecule have revealed that epithelial cells are the predominant cell type expressing transmembrane forms of fractalkine in human skin, the tonsil, and the large intestine. Using these specific anti-fractalkine reagents we do not detect high-level expression of fractalkine on endothelial cells in normal or inflamed colon samples obtained from patients with Crohn's disease or ulcerative colitis. In contrast to previous reports we do not detect fractalkine expression by Langerhans cells or immature dendritic cells in mucosal-associated lymphoid tissues in vivo. We show that the reagent used in previous studies, an anti-fractalkine N-terminal peptide antisera, cross-reacts with human CD84. Finally we discuss potential roles for fractalkine in constitutive leukocyte trafficking based on its observed pattern of expression in epithelia.
Figures

Similar articles
-
Fractalkine is an epithelial and endothelial cell-derived chemoattractant for intraepithelial lymphocytes in the small intestinal mucosa.J Immunol. 2000 Mar 15;164(6):3368-76. doi: 10.4049/jimmunol.164.6.3368. J Immunol. 2000. PMID: 10706732
-
Role for neuronally derived fractalkine in mediating interactions between neurons and CX3CR1-expressing microglia.Proc Natl Acad Sci U S A. 1998 Sep 1;95(18):10896-901. doi: 10.1073/pnas.95.18.10896. Proc Natl Acad Sci U S A. 1998. PMID: 9724801 Free PMC article.
-
Fractalkine, a CX3C chemokine, is expressed by dendritic cells and is up-regulated upon dendritic cell maturation.Eur J Immunol. 1999 Aug;29(8):2551-9. doi: 10.1002/(SICI)1521-4141(199908)29:08<2551::AID-IMMU2551>3.0.CO;2-T. Eur J Immunol. 1999. PMID: 10458770
-
Regulation of CX3CL1/fractalkine expression in endothelial cells.J Atheroscler Thromb. 2004;11(1):15-21. doi: 10.5551/jat.11.15. J Atheroscler Thromb. 2004. PMID: 15067194 Review.
-
[Fractalkine and inflammatory diseases].Nihon Rinsho Meneki Gakkai Kaishi. 2005 Jun;28(3):131-9. doi: 10.2177/jsci.28.131. Nihon Rinsho Meneki Gakkai Kaishi. 2005. PMID: 15997176 Review. Japanese.
Cited by
-
Fractalkine receptor CX(3)CR1 is expressed in epithelial ovarian carcinoma cells and required for motility and adhesion to peritoneal mesothelial cells.Mol Cancer Res. 2012 Jan;10(1):11-24. doi: 10.1158/1541-7786.MCR-11-0256. Epub 2011 Nov 7. Mol Cancer Res. 2012. PMID: 22064656 Free PMC article.
-
Interleukin-10 and interferon-gamma modulate surface expression of fractalkine-receptor (CX(3)CR1) via PI3K in monocytes.Immunology. 2010 Apr;129(4):600-9. doi: 10.1111/j.1365-2567.2009.03181.x. Epub 2009 Sep 9. Immunology. 2010. PMID: 20102414 Free PMC article.
-
Genetic disruption of fractalkine signaling leads to enhanced loss of cochlear afferents following ototoxic or acoustic injury.J Comp Neurol. 2018 Apr 1;526(5):824-835. doi: 10.1002/cne.24369. Epub 2017 Dec 17. J Comp Neurol. 2018. PMID: 29218724 Free PMC article.
-
ESX1-dependent fractalkine mediates chemotaxis and Mycobacterium tuberculosis infection in humans.Tuberculosis (Edinb). 2014 May;94(3):262-70. doi: 10.1016/j.tube.2014.01.004. Epub 2014 Feb 3. Tuberculosis (Edinb). 2014. PMID: 24631198 Free PMC article.
-
Metalloprotease dependent release of placenta derived fractalkine.Mediators Inflamm. 2014;2014:839290. doi: 10.1155/2014/839290. Epub 2014 Mar 13. Mediators Inflamm. 2014. PMID: 24771984 Free PMC article.
References
-
- Bazan JF, Bacon KB, Hardiman G, Wang W, Soo K, Rossi D, Greaves DR, Zlotnik A, Schall TJ: A new class of membrane-bound chemokine with a CX3C motif. Nature 1997, 385:640-644 - PubMed
-
- Pan Y, Lloyd C, Zhou H, Dolich S, Deeds J, Gonzalo JA, Vath J, Gosselin M, Ma J, Dussault B, Woolf E, Alperin G, Culpepper J, Gutierrez-Ramos JC, Gearing D: Neurotactin, a membrane-anchored chemokine upregulated in brain inflammation. Nature 1997, 387:611-617 - PubMed
-
- Combadiere C, Salzwedel K, Smith ED, Tiffany HL, Berger EA, Murphy PM: Identification of CX3CR1. A chemotactic receptor for the human CX3C chemokine fractalkine and a fusion coreceptor for HIV-1. J Biol Chem 1998, 273:23799-23804 - PubMed
-
- Imai T, Hieshima K, Haskell C, Baba M, Nagira M, Nishimura M, Kakizaki M, Takagi S, Nomiyama H, Schall TJ, Yoshie O: Identification and molecular characterization of fractalkine receptor CX3CR1, which mediates both leukocyte migration and adhesion. Cell 1997, 91:521-530 - PubMed
-
- Harrison JK, Jiang Y, Chen S, Xia Y, Maciejewski D, McNamara RK, Streit WJ, Salafranca MN, Adhikari S, Thompson DA, Botti P, Bacon KB, Feng L: Role for neuronally derived fractalkine in mediating interactions between neurons and CX3CR1-expressing microglia. Proc Natl Acad Sci USA 1998, 95:10896-10901 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

