Essential role for cellular phosphoglucomutase in virulence of type 3 Streptococcus pneumoniae
- PMID: 11254588
- PMCID: PMC98160
- DOI: 10.1128/IAI.69.4.2309-2317.2001
Essential role for cellular phosphoglucomutase in virulence of type 3 Streptococcus pneumoniae
Abstract
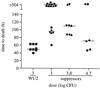
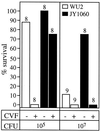
Synthesis of the Streptococcus pneumoniae type 3 capsule requires the pathway glucose-6-phosphate (Glc-6-P) --> Glc-1-P --> UDP-Glc --> UDP-glucuronic acid (UDP-GlcUA) --> (GlcUA-Glc)(n). The UDP-Glc dehydrogenase and synthase necessary for the latter two steps, and essential for capsule production, are encoded by genes (cps3D and cps3S, respectively) located in the type 3 capsule locus. The phosphoglucomutase (PGM) and Glc-1-P uridylyltransferase activities necessary for the first two steps are derived largely through the actions of cellular enzymes. Homologues of these enzymes, encoded by cps3M and cps3U in the type 3 locus, are not required for capsule production. Here, we show that cps3M and cps3U also are not required for mouse virulence. In contrast, nonencapsulated isolates containing defined mutations in cps3D and cps3S were avirulent, as were reduced-capsule isolates containing mutations in pgm. Insertion mutants that lacked PGM activity were avirulent in both immunologically normal (BALB/cByJ) and immunodeficient (CBA/N) mice. In contrast, a mutant (JY1060) with reduced PGM activity was avirulent in the former but had only modestly reduced virulence in the latter. The high virulence in CBA/N mice was not due to the lack of antibodies to phosphocholine but reflected a growth environment distinct from that found in BALB/cByJ mice. The reduced PGM activity of JY1060 resulted in enhanced binding of complement and antibodies to surface antigens. However, decomplementation of BALB/cByJ mice did not enhance the virulence of this mutant. Suppressor mutations, only some of which resulted in increased capsule production, increased the virulence of JY1060 in BALB/cByJ mice. The results suggest that PGM plays a critical role in pneumococcal virulence by affecting multiple cellular pathways.
Figures




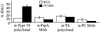
References
-
- Arrecubieta C, Garcia E, Lopez R. Sequence and transcriptional analysis of a DNA region involved in the production of capsular polysaccharide in Streptococcus pneumoniae type 3. Gene. 1995;167:1–7. - PubMed
-
- Ausubel F M, Brent R, Kingston R E, Moore D D, Seidman J G, Smith J A, Struhl K, editors. Current protocols in molecular biology. New York, N.Y: John Wiley and Sons, Inc.; 1994.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

