Mitogenic effect of oxidized low-density lipoprotein on vascular smooth muscle cells mediated by activation of Ras/Raf/MEK/MAPK pathway
- PMID: 11264247
- PMCID: PMC1572708
- DOI: 10.1038/sj.bjp.0703976
Mitogenic effect of oxidized low-density lipoprotein on vascular smooth muscle cells mediated by activation of Ras/Raf/MEK/MAPK pathway
Abstract
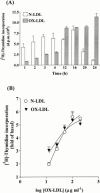
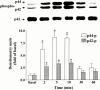
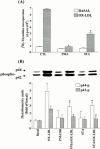
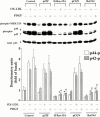
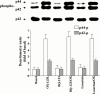
1. It has been demonstrated that oxidized low-density lipoprotein (OX-LDL) is a risk factor in atherosclerosis by stimulating vascular smooth muscle cell (VSMC) proliferation. However, the mechanisms of OX-LDL-induced cell proliferation are not completely understood. Therefore, we investigated the effect of OX-LDL on cell proliferation associated with mitogen-activated protein kinase (MAPK) activation in rat cultured VSMCs. 2. Both native-LDL (N-LDL) and OX-LDL induced a time- and concentration-dependent incorporation of [(3)H]-thymidine in VSMCs. 3. OX-LDL induced time- and concentration-dependent phosphorylation of p42/p44 MAPK. Pretreatment of these cells with pertussis toxin or U73122 attenuated the OX-LDL-induced responses. 4. Pretreatment with PMA for 24 h, preincubation with a PKC inhibitor staurosporine or the tyrosine kinase inhibitors, genistein and herbimycin A for 1 h, substantially reduced [(3)H]-thymidine incorporation and p42/p44 MAPK phosphorylation induced by OX-LDL. 5. Removal of Ca(2+) by BAPTA/AM or depletion of the internal Ca(2+) pool by thapsigargin significantly inhibited OX-LDL-induced [(3)H]-thymidine incorporation and p42/p44 MAPK phosphorylation. 6. OX-LDL-induced [(3)H]-thymidine incorporation and p42/p44 MAPK phosphorylation was inhibited by PD98059 (an inhibitor of MEK1/2) and SB203580 (an inhibitor of p38 MAPK) in a concentration-dependent manner. 7. Overexpression of dominant negative mutants of Ras (H-Ras-15A) and Raf (Raf-N4) significantly suppressed MEK1/2 and p42/p44 MAPK activation induced by OX-LDL and PDGF-BB, indicating that Ras and Raf may be required for activation of these kinases. 8. These results suggest that the mitogenic effect of OX-LDL is mediated through a PTX-sensitive G protein-coupled receptor that involves the activation of the Ras/Raf/MEK/MAPK pathway similar to that of PDGF-BB in rat cultured VSMCs.
Figures






References
-
- AUTIO I., JAAKOLA O., SOLAKIVI T., NIKKARI T. Oxidized low-density lipoprotein is chemotactic for arterial smooth muscle cells in culture. FEBS Lett. 1990;277:247–249. - PubMed
-
- BOULANGER C.M., TANNER F.C., B'EA M.L., HAHN A.W., WERNER A., LUSCHER T.F. Oxidized low density lipoproteins induce mRNA expression and release of endothelin from human and porcine endothelium. Cir. Res. 1992;70:1191–1197. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

