Propagation of intercellular calcium waves in retinal astrocytes and Müller cells
- PMID: 11264297
- PMCID: PMC2409971
- DOI: 10.1523/JNEUROSCI.21-07-02215.2001
Propagation of intercellular calcium waves in retinal astrocytes and Müller cells
Abstract
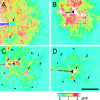
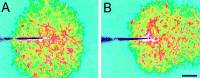
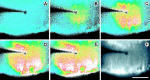
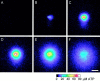
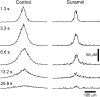
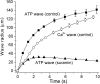
Intercellular Ca(2+) waves are believed to propagate through networks of glial cells in culture in one of two ways: by diffusion of IP(3) between cells through gap junctions or by release of ATP, which functions as an extracellular messenger. Experiments were conducted to determine the mechanism of Ca(2+) wave propagation between glial cells in an intact CNS tissue. Calcium waves were imaged in the acutely isolated rat retina with the Ca(2+) indicator dye fluo-4. Mechanical stimulation of astrocyte somata evoked Ca(2+) waves that propagated through both astrocytes and Müller cells. Octanol (0.5 mm), which blocks coupling between astrocytes and Müller cells, did not reduce propagation into Müller cells. Purinergic receptor antagonists suramin (100 microm), PPADS (20-50 microm), and apyrase (80 U/ml), in contrast, substantially reduced wave propagation into Müller cells (wave radii reduced to 16-61% of control). Suramin also reduced wave propagation from Müller cell to Müller cell (51% of control). Purinergic antagonists reduced wave propagation through astrocytes to a lesser extent (64-81% of control). Mechanical stimulation evoked the release of ATP, imaged with the luciferin-luciferase bioluminescence assay. Peak ATP concentration at the surface of the retina averaged 78 microm at the stimulation site and 6.8 microm at a distance of 100 microm. ATP release propagated outward from the stimulation site with a velocity of 41 microm/sec, somewhat faster than the 28 microm/sec velocity of Ca(2+) waves. Ejection of 3 microm ATP onto the retinal surface evoked propagated glial Ca(2+) waves. Together, these results indicate that Ca(2+) waves are propagated through retinal glial cells by two mechanisms. Waves are propagated through astrocytes principally by diffusion of an internal messenger, whereas waves are propagated from astrocytes to Müller cells and from Müller cells to other Müller cells primarily by the release of ATP.
Figures


References
-
- Araque A, Parpura V, Sanzgiri RP, Haydon PG. Glutamate-dependent astrocyte modulation of synaptic transmission between cultured hippocampal neurons. Eur J Neurosci. 1998a;10:2129–2142. - PubMed
-
- Bezzi P, Carmignoto G, Pasti L, Vesce S, Rossi D, Lodi Rizzini B, Pozzan T, Volterra A. Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature. 1998;391:281–285. - PubMed
-
- Boitano S, Dirksen ER. Intercellular propagation of calcium waves mediated by inositol trisphosphate. Science. 1992;258:292–295. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
